Received: February 2026
DOI 10.17677/fn20714807.2026.01.02
Fluorine Notes, 2026, 164, 3-4
THE STRUCTURE OF PARAMAGNETIC CENTERS AND THE ANALYSIS OF THE ELEMENTARY STAGES OF THE MECHANISM OF THE STABILIZING EFFECT OF FLUORINATED TETRAAMINE ON THE OZONE AGING PROCESS OF A MODIFIED POLYURETHANE ELASTOMER
I.A. Politsimako, S.V. Kudashev, V.F. Zheltobryukhov, and S.V. Kumbrasyeva
Volgograd State Technical University, 28 Lenin Avenue, Volgograd, 400005 Russia
e-mail: kudashev-sv@yandex.ru
Abstract. The modifying effect of fluorinated tetraamine synthesized by the interaction of tris-(2-aminoethylamine) and 1H,1H,3H-trihydroperfluoropropane-1-ol on the properties of a modified polyurethane elastomer was studied. The stabilizing effect of the fluorinated modifier used on the properties of the elastomer under the influence of an ozone-air mixture is shown. The structure of paramagnetic centers and the possible mechanism of binding of ozone molecules are considered.
Keywords: fluoropolymers; polyurethane elastomers; fluorinated amines; modification; stabilization; ozone aging; paramagnetic centers.
Introduction
The operation of polyurethane sports, roofing, and waterproofing coatings causes multifactorial processes of macromolecule destruction of the heterochain polymer under consideration [1,2]. The destructive effects of aggressive media, UV radiation, and changing air temperatures can be reduced by modifying polyurethane materials with poly- and perfluorinated compounds [3-6].
The use of indifferent additives (fluoroparaffins, polytetrafluoroethylene, fluorinated inorganic compounds) leads to their partial migration from the polymer volume during the operation of coatings [3]. The introduction of fluorinated reactive compounds (isocyanates, alcohols, carboxylic acids, amines, and multifunctional compounds) allows chemical modification of polyurethanes [7,8] by «embedding» fluorinated fragments into macromolecules and eliminating possible diffusion of the modifier from the polymer during coating operation.
The combination of amino groups of varying degrees of substitution and polyfluorinated fragments in the structure of the modifier, which can collectively participate in the disruption of free radical processes occurring under the influence of an ozone-air mixture, opens up new prospects for stabilizing the properties of polyurethane materials.
The aim of the work is to identify the structural features of paramagnetic centers formed when exposed to an ozone-air mixture on a polyurethane elastomer modified with the bisalkylation product tris-(2-aminoethylamine) 1H,1H,3H-trihydroperfluoropropane-1-ol.
Experimental part
The basic polyurethane elastomer was obtained using a laboratory mixer by mixing (mixing speed 250 rpm-1) for 10 min. 100 wt.h. oligoesterpoliol (Laprol 5003-2–B10 (hydroxyl number 35 mg KOH/g, mass fraction of water not more than 0.05%, Jiahua Chemical Co., LTD), 1 wt.h. of the branching chain agent (glycerin clean for analysis, JSC «EKOS-1»), 1.5 wt.h. of the plasticizer (dioctyladipinate DOA, the content of the basic substance 99.7% (wt.), LLC «Vitakhim SPb»), 1.5 wt.h. surfactant (oxyethylated monoalkylphenol Neonol AF 9-12, mass fraction of water < 0.5%, NPC PROMKHIMPLAST LLC), 0.1 wt.h. of urethane catalyst (2.5% solution of di-n-butyldilaurate of tin in white spirit, PTK Neftepromkomplekt LLC). Next, 20 wt.h. of isocyanate (Desmodur T80, 2,4-isomer content 80.5%, «Wanhua») was added to the reaction mass and mixed again for 7 minutes. The [NCO] / [OH] in the formulation of the composition was 1.50 / 1.00.
A polyurethane elastomer modified with fluorinated tetraamine was obtained similarly to the method described above by introducing the specified modifier in an amount of 5 wt.h. into the reaction mass at the stage of mixing oligoesterpoliol, glycerol, dioctyladipinate, oxyethylated monoalkylphenol and a urethane catalyst.
Fluorinated tetraamine was obtained according to scheme (I) by catalytic N-polyfluoroalkylation of tris-(2-aminoethyl)amine (tkip = 114 °C (15 mm Hg), d = 0.976 g/ml, n20D = 1.497, «Keyingchem») 1H,1H,3H-trihydroperfluoropropane-1-ol (basic substance content 99.5% (wt.), JSC «HaloPolymer») in the presence of catalytic amounts of montmorillonite (base substance content 99% (wt.), TOO «B-Clay») in a sealed glass ampoule at 80 ° C for 2 hours at an ultrasound frequency of 40 kHz, followed by heating to 120 ° C for 6 hours. Fluorinated tetraamine was a yellow oily substance with a temperature of = 131-133 °C (15 mmHg).
Scheme (I)
The obtained compositions of the reference and modified polyurethane elastomer were poured into molds and kept at room temperature (cold curing method) until the Shore A hardness of the elastomer reached a plateau.
Ozone aging of polymer samples was carried out under static conditions under conditions of continuous supply of an ozone-air mixture generated by an Ozon–6000 ozonizer (100 W, capacity 60 g/h). The distance from the ozonator to the sample (polyurethane coating on a concrete substrate) was 35 cm. Test conditions: 8 h (30 min exposure / 60 min rest) – exposure of samples at a temperature of 23 ± 3 ° C, 16 h – rest. The shape and dimensions of the samples corresponded to type 2 according to GOST 21751-76.
The wetting edge angle was determined using an OCA 15 EC device from DataPhysics with integrated SCA 20 software. The measurements were carried out by applying drops of test liquid (deionized water (AO Lenreactive) with a volume of 5-7 µl to the surface of the elastomer and calculating the wetting angle of the sedentary droplet using the Young–Laplace method. 6-8 parallel measurements were performed and the arithmetic mean of the edge angles was calculated.
Quantum chemical studies in the approximation of an isolated particle in the gas phase with geometry optimization in all parameters using the DFT–PBE0/6–311g** and ab initio methods based on STO–3G** were performed in the GAMESS and Gaussian 09 software products.
Discussion of the results
Aromatic amines, spatially hindered phenols, wax, dialkyldithiocarbamates of metals of variable valence, derivatives of urea and thiourea, diphenyl sulfide, and benzofuran have been described in the literature as antiozonants [1, 2, 9-12]. As a rule, with a low content of the antiozonant, not exceeding 2-3 wt.h. per 100 wt.h. of rubber, undesirable staining of the elastomer is already observed.
The process of migration polymerization of isocyanate and polyol in the presence of fluorinated tetraamine, leading to the formation of modified polyurethane, is accompanied by the involvement of amino groups of the modifier in reaction with diisocyanate according to scheme (II). According to combined data from [13] 1H NMR and X–ray photoelectron spectroscopy, the proportion of groups containing unsubstituted nitrogen –NH-CH2CF2 in the cured modified polyurethane elastomer is no more than 24-26% by weight. it depends on the theoretically possible, and depends on the number of units of –CF2–CF2– in the composition of –NH–CH2(CF2CF2)nH. The remaining groups undergo interaction with 2.4- and 2.6-toluene diisocyanates to form disubstituted urea according to scheme (II) during the structure formation of the crosslinked polymer.
Scheme (II)
The secondary amino groups in the composition –NH–CH2CF2 are characterized by lower reactivity due to the electron acceptor effect of fluorine and steric difficulties in the course of reactions with their participation. The structural fragment >N–CH2CH2–NH–CH2CF2CF2H in the resulting modified polymer is capable of intra- and intermolecular non-covalent interactions in the macromolecular system (N–H···O=C, N–H···*O<, N–H···F–C, C–H···F–C), changing the segmental mobility of the chains, the proportion of physical connections and forming a more densely packed grid. Another feature is the possibility of generating paramagnetic centers of various chemical structures according to scheme (III) when ozone molecules interact with fluorinated and amino groups that are part of polyurethane urea macromolecules.
At the first stage, the mutual orientation of the reacting molecules and their dipole-dipole interactions occur (the angle between the bonds in the O3 molecule is 116.8°). According to the DFT-PBE0/6–311g** method, the high electron density on the oxygen atom of the O3 molecule and the positive charge on the hydrogen atoms of CF2H+0.080 and N–0.112–H+0.247 (1a) groups favor the further formation of an ion pair (1b). In this case, the O–O bond lengthens from 1.278 Å to 1.406 Å, which then leads to the formation of a single N–O bond with a length of 1.431 Å. The activation barrier of the transition (1a) (1b) is 41 kcal/mol. An increase in the magnitude of the dipole moment is observed from 5.30 D (1a) to 8.10 D (1b).
The elimination of singlet oxygen (through the stage of generation of the ion pair (1c) +N–O-) promotes the formation of the final product, hydroxylamines (1d). The formation of >N–OH can proceed through the generation of aminyl radicals >N• (2b), the subsequent interaction of which with alkoxy radicals will lead to nitroxyl esters >N–OR, and with the hydroperoxide radical, the product (2c). The transition (1b) (2a) is characterized by an energy barrier of 10 kcal/mol. Further cleavage of the proton from the radical cation (2a) promotes the formation of an aminyl radical. The stabilization of the (2a) structure is influenced by the non-covalent interactions between the ozonide anion and the proton of the HCF2 group.
The activation barrier of the (1b) (3a) transition is 9.5 kcal/mol and is comparable to the barrier of formation of the radical cation (1b) (2a). The elimination of the hydroperoxide radical from the ion pair (3a) is accompanied by the formation of a nitroxyl radical (3d). According to the ab initio method (STO–3G**), the approximate ratio of ion pairs (3b) and (3c) is 3:1. The dipole moments are 7.15 D (3b) and 4.00 D (3c), and the activation barrier of the transition (3b) (3c) has a significant value equal to 83 kcal/mol. The bond length +N–O● ··· -CF2 is 0.960 Å, while +N–H+δ ··· -CF2 is 2.112 Å.
A study of the surface properties of polyurethane elastomers has shown that the introduction of fluorinated tetraamine promotes the hydrophobization of modified polyurethane (Table). The most significant change in the value of the marginal wetting angle of the surface, after ozone aging, is characteristic of the initial polyurethane, due to the formation of oxidation products. For a modified polyurethane elastomer containing ozone–resistant groups –CF2–CF2–, the changes in the edge angle are not so significant.
Table 1. The marginal wetting angle of the surface of polyurethane elastomers
|
Sample |
Marginal wetting angle, ° |
|
|
before ozone aging |
after ozone aging |
|
|
The initial polyurethane elastomer |
89±2 |
73±2 |
|
Modified polyurethane elastomer containing fluorinated tetraamine |
106±1 |
98±2 |
Thus, increasing the stability of polyurethane elastomers modified with tris-(2-aminoethyl) bisalkylation productamine 1H,1H,3H-trihydroperfluoropropane-1-ol, under the influence of an ozone-air mixture, the combined participation of HCF2CF2 groups, nitroxyl and aminyl free radicals in the binding of paramagnetic centers formed during the decay of macromolecular chains contributes.
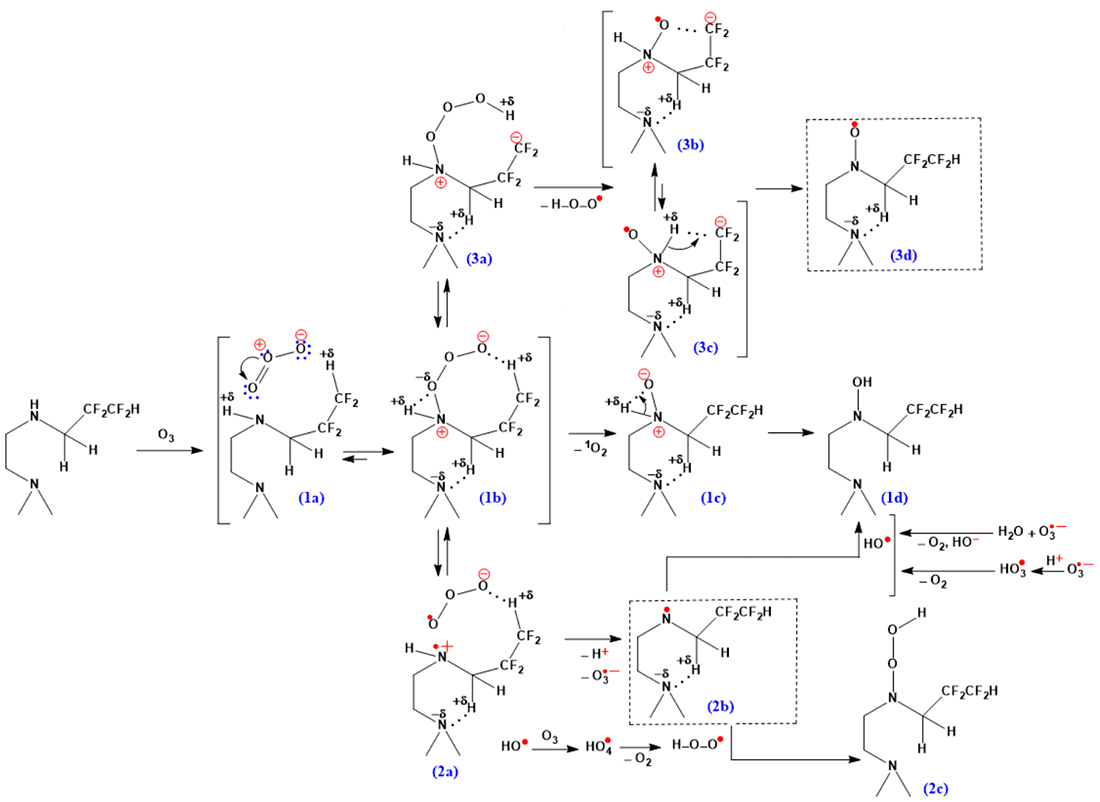
Scheme (III)
References
- Thomas S., Datta J., Haponiuk J. Polyurethane polymers: Composites and nanocomposites. Elsevier, Amsterdam, Netherlands, 2017. 632 р.
- Clemitson I. R. Castable Polyurethane Elastomers. – CRC Press (Taylor & Francis Group), 2015. 272 p.
- Ebnesajjad S., Morgan R. A. Fluoropolymer Additives. Elsevier, William Andrew, 2019. P. 57–66.
- Smirnova O., Glazkov A., Yarosh A., Sakharov A. Fluorinated Polyurethanes, Synthesis and Properties // Molecules. 2016. V. 21. N 7. Р. 1–10. https://doi.org/10.3390/molecules21070904
- Wu Z., Tang L., Dai J., Qu J. Synthesis and properties of fluorinated non-isocyanate polyurethanes coatings with good hydrophobic and oleophobic properties // J. Coat. Technol. Res. 2019. V. 16. Р.1233–1241. http://dx.doi.org/10.1007/s11998-019-00195-5
- Li N., Yang R., Tian Y., Lu P., Huang N., Li H., Chen X. Synthesis of durable hydrophobic fluorinated polyurethanes with exceptional cavitation erosion resistance // Tribol. Int. 2023. V. 177. ID. 107973. https://doi.org/10.1016/j.triboint.2022.107973
- Ponomarenko V.A., Krukovskij S.P., Aly`bina A.Yu. Ftorsoderzhashhie geterocepny`e polimery`. – M.: Nauka, 1973. 304 p.
- Malichenko B. F. Ftorsoderzhashhie poliamidy` i poliuretany`. – Kiev: Naukova dumka. 1977. 231p.
- Neiman M. B. B. Aging and Stabilization of Polymers (1965th Edition). Springer, 2012. 376 р.
- Razumovskij S. D., Zaikov G. E. Ozon i ego reakcii s organicheskimi soedineniyami. – M.: Nauka, 1974. 322 s.
- Zaikov G., Rakovsky S. Ozonation of Organic and Polymer Compounds. – Smithers Rapra, Shawbury, 2009. 416 p.
- Kudashev S.V., Medvedev V.P., Tuzhikov O.O. Study of Ozone Aging of Fluorine-Containing Polydienurethane Elastomers // Protection of Metals and Physical Chemistry of Surfaces. 2019. Vol. 55, No. 2. P. 359-362. https://doi.org/10.1134/S2070205119020138.
- Kudashev S.V., Shulenina A.V., Peters G.S., Policimako I.A., Klimov V.V., Zheltobryuxov V.F. Fotoximicheskaya destrukciya i agressivostojkost` ftorsoderzhashhix modificirovannyx poliuretanovyx elastomerov // Fizikoximiya poverxnosti i zashhita materialov. 2025. V. 61, N 4. P. 432-441. https://doi.org/10.31857/S0044185625040119.
ARTICLE INFO
Received 18 February 2026
Accepted 27 February 2026
Available
online February 2026
Recommended for publication by PhD O.V. Bryzgalova
eLIBRARY Document Number (EDN) OJVODJ

Fluorine Notes, 2026, 164, 3-4


