Received: September 2022
DOI 10.17677/fn20714807.2022.05.02
Fluorine Notes, 2022, 144, 3-4
INVESTIGATION OF OPTICAL PROPERTIES OF AMORPHOUS COPOLYMERS OF PERFLUORO-2,2-DIMETHYL-1,3-DIOXOL AND PERFLUORO-(2-CYCLOPENTYL) ETHYLVINYL ETHER OBTAINED AT ULTRAHIGH PRESSURE
V, I. Sokolov1,2, I.O. Goryachuk1, S.I. Molchanova1, E.V. Polunin3
1Federal Scientific Research Center "Crystallography and Photonics" RAS, 119333, Leninsky ave. 59, Moscow, Russia
2Federal Scientific Center, Scientific Research Institute for System Research of RAS, 117218, Nakhimovsky ave. 36, bldg. 1, Moscow, Russia
3N. D. Zelinsky Institute of Organic Chemistry Russian Academy of Sciences, 119991, Leninsky ave. 47, Moscow, Russia
Abstract: Copolymers of perfluoro-2,2-dimethyl-1,3-dioxol and perfluoro-(2-cyclopentyl)-ethylvinyl ether with different molar content of ether units in the macromolecule have been synthesized by the ultrahigh pressure method without using of initiators. The resulting copolymers have high optical transparency in visible and near IR spectral ranges and low refractive index n=1.293-1.336 (at a wavelength of λ=632.8 nm), which is determined by the ratio of dioxolane and ether units. The synthesized copolymers are amorphous, capable of film formation, and can be used to create various waveguide elements of integrated optical devices.
Keywords: Amorphous perfluorinated copolymers, dioxols, vinyl ethers, ultrahigh pressure polymerization, refractive index, light guiding films, optical waveguides.
Introduction
Amorphous perfluorinated polymers with high optical transparency and low refractive index n within “telecommunication” wavelength ranges λ near 0.85, 1.3 and 1.55 µm are promising for creating the optical waveguides and other elements of integrated optical devices [1-4]. Unlike their hydrocarbon analogues the perfluoropolymers have high chemical resistance and are more resistant to elevated temperatures (less prone to yellowing), since C-F bond in the polymer macromolecule is stronger than C-H bond.
The product line of commercially available amorphous perfluorinated polymers currently is very limited. To the best of our knowledge, such polymers are commercially produced only by DuPont company (TeflonAF-type perfluoropolymers), Asachi Glass company (Cytop perfluoropolymer) and Solvay company (Hyflon AD) [5]. Analysis of US and European patents [6-12] shows that synthesis of new amorphous perfluorinated polymeric materials with improved set of useful properties is an urgent task.
Earlier, using the method of ultrahigh pressure (12-16 ths. atm.) at a temperature of 100-150°С, we synthesized a number of amorphous copolymers of perfluorinated dioxols and vinyl ethers [13-15], which under normal conditions with great difficulties enter into a radical polymerization reaction. In this paper, we report about synthesis of amorphous copolymers of perfluoro-2,2-dimethyl-1,3-dioxol and perfluoro-(2-cyclopentyl)ethyl vinyl ether with different molar content of ether units in the macromolecule using ultrahigh pressure method. The resulting copolymers have a high optical transparency in visible and near-IR spectral regions, as well as a low refractive index n = 1.293–1.336 (at a wavelength of λ= 632.8 nm), which is determined by the ratio of dioxolane and ether units. These copolymers are capable to film formation and can be used to create various waveguide elements of integrated optical devices.
Synthesis of amorphous perfluorinated copolymers at ultrahigh pressure
The monomers perfluoro-2,2-dimethyl-1,3-dioxol D1 and perfluoro-(2-cyclopentyl)-ethylvinyl ether EC5 were used to prepare the copolymers (see Fig. 1). These monomers are transparent colorless liquids, the degree of their chemical purity was more than 99%. Synthesis of copolymers was carried out without using of initiators in Teflon ampoules with a volume of 2-3 ml in press molds of cylinder–piston type at a pressure of 15-16 ths. atm. and temperature 155-165°C. The reaction time varied from 96 to 192 h. Prior to synthesis, the monomers were distilled in argon atmosphere to remove dissolved oxygen, which is known to be an inhibitor of radical polymerization reaction. The product obtained after opening the ampoule was, as a rule, a highly viscous transparent substance containing, in addition to linear copolymer, the highly volatile components (dimers, oligomers and other reaction by-products). To remove these components, the copolymers were vacuum-processed to constant weight at 120°C.
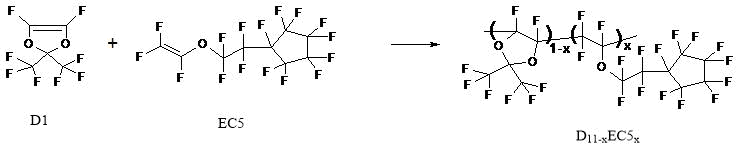
Figure 1. Synthesis scheme of copolymers of perfluoro-2,2-dimethyl-1,3-dioxol D1 and perfluoro-(2-cyclopentyl) ethylvinyl ether EC5 by radical polymerization at ultrahigh pressure without using of initiators. x is the molar concentration of ether units in D11-xEC5x copolymer macromolecule.
Described method was used to synthesize of D11-xEC5x copolymers with different molar content x of ether units in macromolecules. Fig. 2a shows 19F NMR spectrum of EC5 monomer, and Fig. 2b - copolymer D11-xEC5x with x = 0.5 (b). These spectra were measured in perfluorobenzene via Bruker AM-300 spectrometer (at 282.40 MHz).
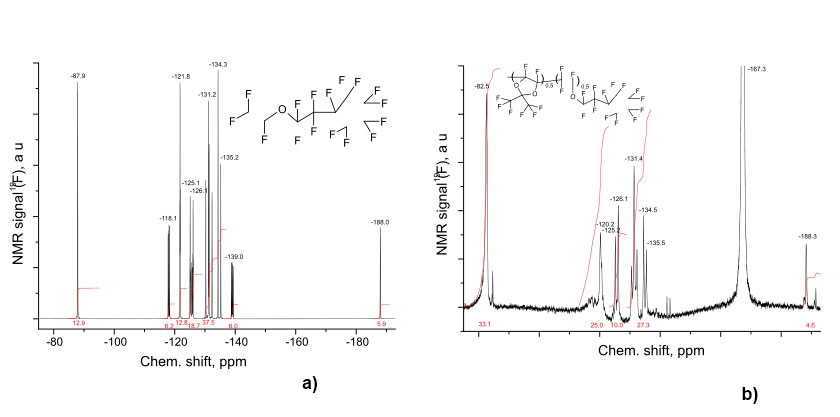
Figure 2. 19F NMR spectra of EC5 monomer (a) and D11-xEC5x copolymer with x = 0.5 (b), measured via Bruker AM-300 spectrometer (at 282.40 MHz).
Broadened singlet at -82.6 ppm in Fig. 2b corresponds to six fluorine atoms in the methyl groups of perfluorodioxol D1; CF2O signal from side chain of polymer was superimposed on it. The multiplet at -120 ppm (2F) is the signal from CF2 group adjacent to the cycle. The multiplets at -125 (2F), -131 (4F), and -135 ppm correspond to CF2 groups of the cycle. In the same range from -110 to -140 ppm broadened signals from fluorine atoms are located in the main chain of polymer. Fig. 2a and Fig. 2b shows the signal -188 ppm corresponds to CF group in the cyclopentyl fragment of ether EC5, and signal at -167.4 ppm associated with solvent (hexafluorobenzene). The ratio of dioxolane and ether fragments in the polymer chain was determined by comparing the integral intensities of signals described above for CF2 and CF3 groups. From the analysis of Fig. 2b, we can conclude that the ratio of dioxol and ether units in the copolymer is D1:EC5 1:1 (x = 0.5).
To estimate the molecular weight of D11-xEC5x copolymers, the average hydrodynamic diameter of macromolecular globules in perfluorooctane was measured by dynamic light scattering method using 90Plus_Zeta particle/protein analyzer (Brookhaven Instruments Corp., USA) under illumination with laser beam with a wavelength of 640 nm. Fig. 3 shows a typical histogram of globule size distribution. It can be seen that average globules diameter is <D> = 36 nm. Thus, D11-xEC5x copolymers synthesized by radical copolymerization at ultrahigh pressure without using of initiators can be attributed to the class of macromolecular substances.
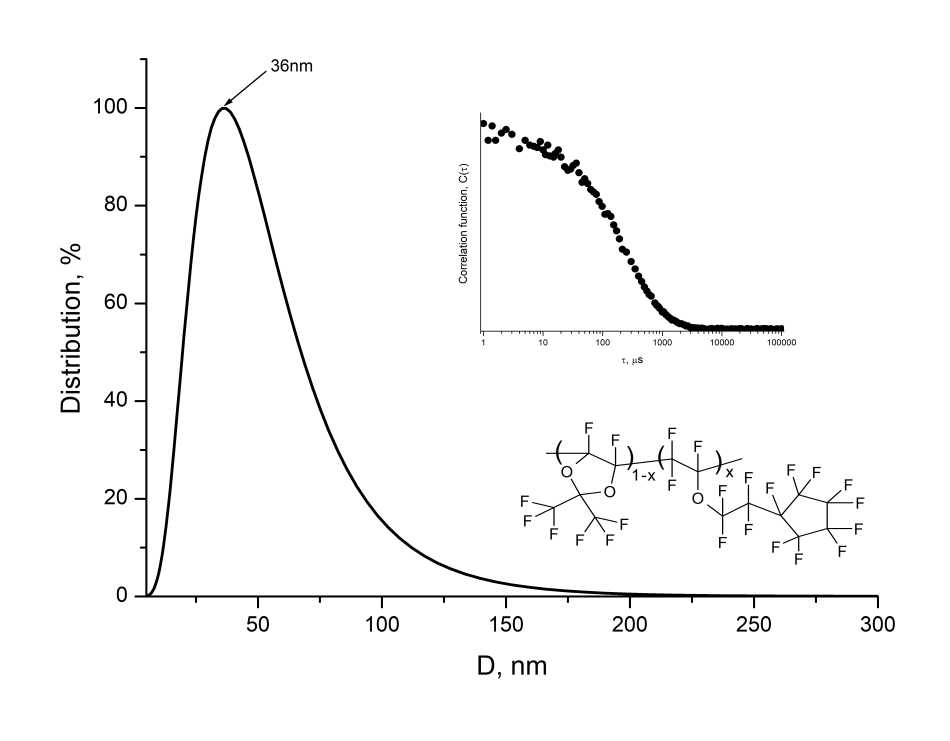
Figure 3. Distribution histogram of macromolecular globules of D11-xEC5x copolymer with х=0.75 by size, measured by dynamic light scattering method in perfluorooctane, where D is globule diameter. The insets show the form of autocorrelation function and the fragment of copolymer structure.
Synthesized copolymers do not dissolve in acetone, hexane, octane, benzene, toluene, but dissolve at room temperature in perfluorinated solvents such as perfluorohexane, perfluorooctane, hexafluorobenzene, perfluoro-(1,3-dimethyl)-cyclohexane (carbogal), perfluorodecalin.
Investigation of perfluorinated copolymers structure by wide angle X-ray scattering
Synthesis of copolymers at ultrahigh pressure in the absence of any initiator occurs according to radical mechanism [13-15]. In this case, the formation of ordered structure capable of leading to appearance of polycrystallites in the copolymer and, accordingly, to increase in light scattering, is unlikely for the following reasons. First, the interlocking of EC5 ether units in the polymeric macromolecule can be either “head-to-head” or “head-to-tail”. Second, ether and dioxolane can occur in different sequences in the polymer chain. In addition, all asymmetric carbon atoms can have either R- or S-configuration. These factors increase the degree of disorder of units in the macromolecule, which leads, as will be shown below, to formation of amorphous materials.
Structural diagnostics of all copolymers was carried out via Rigaku Miniflex600 wide-angle X-ray diffractometer (Cu, λ=1.54184 Å). For this, some films were made from copolymers on glass substrates. The diffraction pattern of film from D11-xEC5x with x=0.25 is shown in Fig. 4. As follows from Fig. 4, this diffraction pattern have not sharp peaks, but only wide “halos” are observed near 2θ≈ 15.2 and 39.5 deg, which indicates the amorphism of this polymer material.
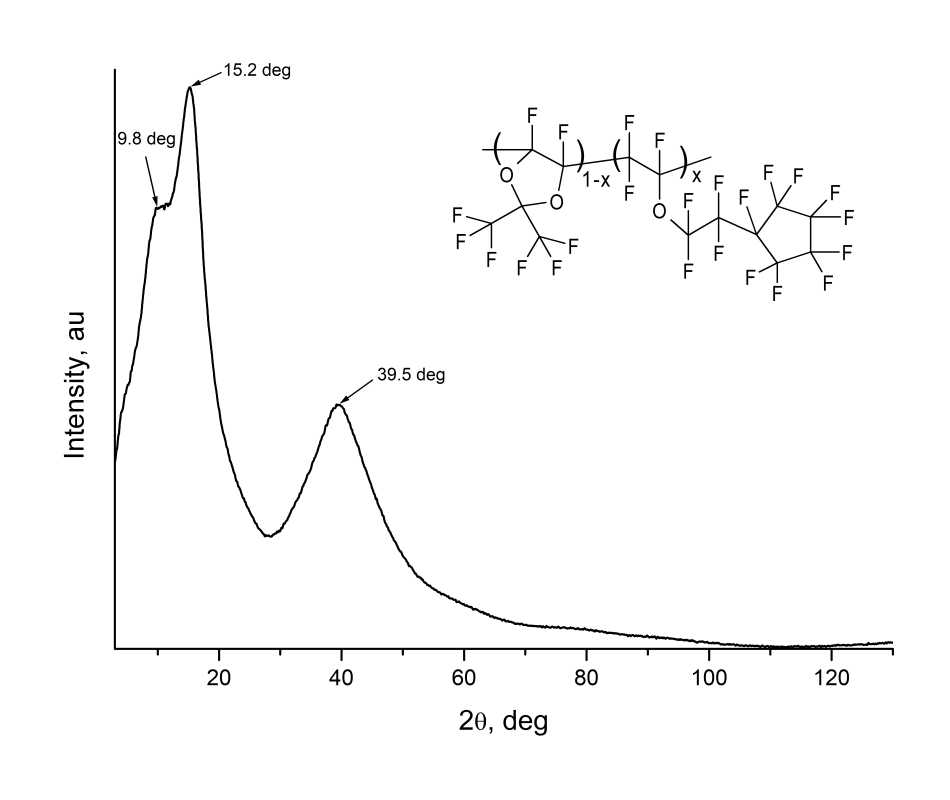
Figure 4. Obtained via Rigaku Miniflex600 X-ray diffractometer X-ray diffraction pattern of film made from D11-xEC5x copolymer with x = 0.25 r.
Investigation of IR absorption spectra of perfluorinated copolymers
The IR absorption spectra of copolymers were measured via Shimadzu8400 FTIR spectrometer. For this, polymer films 1–10 µm thick were formed by centrifugation at KBr substrates. The transmission spectra of clean substrate and of substrate with a polymer film deposited on it were measured. From their ratio, the absorption spectrum of these copolymers was determined. Figure 5 shows the absorption spectrum of the film made from D11-xEC5x perfluorinated copolymer with x = 0.5.
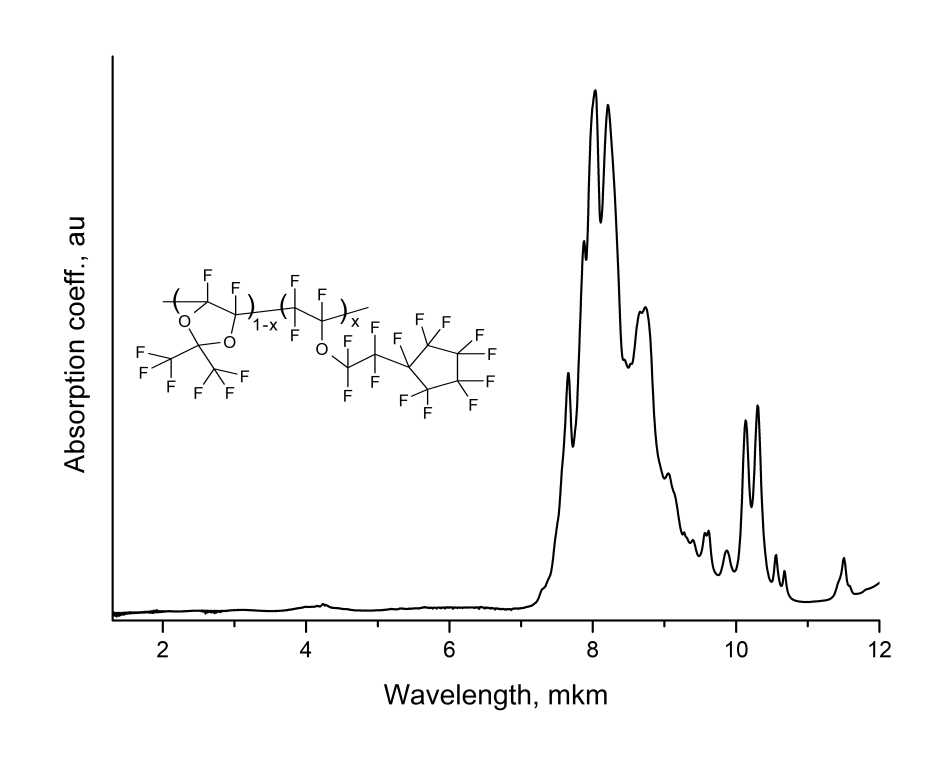
Figure 5. Absorption spectrum of a film made of amorphous perfluorinated copolymer D11-xEC5x with x = 0.5 deposited to KBr substrate by centrifugation method.
As can be seen from Fig. 5, this copolymer does not have intense absorption bands below 2 µm, which indicates its high optical transparency within “telecommunication” C-wavelength range of 1530–1565 nm. The most intense absorption bands of this copolymer lie within the range of 7.5-10 μm and are due to stretching vibrations of CF, CF2 and CF3 groups in the macromolecule.
Measurement of refractive index of perfluorinated copolymer by refractometric method
The study of refractive index n of light-guiding films made from D11-xEC5x copolymers at KBr substrates, as well as at silicon substrates with thermally grown oxide layer, was carried out at a wavelength of λ= 632.8 nm by refractometric method using a Metricon2010/M prism coupling refractometer. The measurements performed at TE and TM polarizations of probing laser beam showed that these films are isotropic (the values of refractive indices nTE, nTM in the directions along and across the film are close), and n increases with increasing molar concentration x of units of perfluorinated ether EC5 in the macromolecule (see Fig. 6). As can be seen from Fig. 6, the refractive index of EC5 ether homopolymer is n = 1.3359 ± 0.0005.
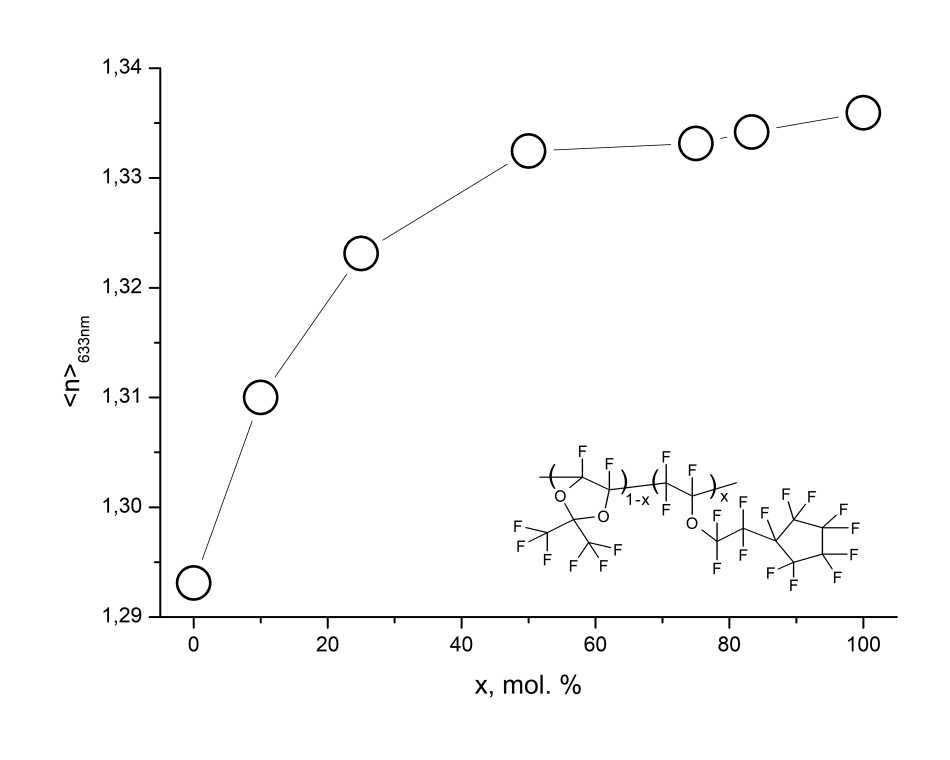
Figure 6. Average refractive index n = (nTE + nTM)/2 of D11-xEC5x copolymers at a wavelength of 632.8 nm as a function of molar concentration x of ether units in the macromolecule, measured using Metricon2010/M prism coupler.
Thus, amorphous perfluorinated copolymers D11-xEC5x are capable to film formation and can be used to form waveguides and other elements of integrated optical devices. In particular, these copolymers can be used as waveguide cladding for manufacture of optical data transmission buses at the printed circuit boards. The low values of refractive index of these copolymers make it possible to use them also as a sheath of high-aperture quartz fibers.
Conclusion
Amorphous copolymers of perfluoro-2,2-dimethyl-1,3-dioxol and perfluoro-(2-cyclopentyl)ethylvinyl ether have been synthesized by ultrahigh pressure method. These copolymers have high optical transparency within “telecommunication” wavelength ranges near 0.85, 1.3, and 1.5 μm, have a low refractive index, are capable of film formation and are promising for manufacture of various waveguide elements of integrated optical devices. This expands the class of amorphous perfluorinated polymers for various applications in photonics. The synthesized copolymers can be used, among other things, as a shell for manufacture of waveguide amplifiers for “telecommunication” C-wavelength range of 1530–1565 nm based on polymers with embedded erbium-doped nanocrystals.
Acknowledgement
This work was financially supported by the Ministry of Science and Higher Education of the Russian Federation within the framework of the state task of the Federal Research Center “Crystallography and Photonics” of the Russian Academy of Sciences in terms of the synthesis of amorphous perfluorinated polymers at ultrahigh pressure and by the Russian Foundation for Basic Research (grant no. 20-07-01038) in terms of studying the refraction of perfluoropolymers. The equipment of the Center for Collective Use of the Federal Research Center “Crystallography and Photonics” of the Russian Academy of Sciences was used in the work.
References
1. Groh W., Overtone absorption in macromolecules for polymer optical fibers, Makromol. Chem., 1988, 189, 2861-2874.
2. Groh W., Zimmermann A., What is the lowest refractive index of organic polymers, Macromolecules, 1991, 24, 6660-6663.
3. Zhou M., Low-loss polymeric materials for passive waveguide components in fiber optical telecommunication., Opt. Eng., 2002, 41(7), 1631-1643.
4. Vitale A., Bongiovanni R., Ameduri B., Fluorinated oligomers and polymers in photopolymerization, Chemical Reviews, 2015, 115(16), 8835-8866.
5. Ameduri B., The promising future of fluoropolymers, Macromolecular Chemistry and Physics, Wiley-VCH Verlag, 2020, 221(8), 1900573.
6. Resnick P. R., Polymers of fluorinated dioxoles, US Patent 3978030, Aug. 31, 1976.
7. Imbalzano, J. F.,. Kerbow D.L., Stable tetrafluoroethylene copolymers, US patent 4743658, May 10, 1988.
8. Colaianna P., Brinati G., Arcella V., Amorphous perfluoropolymers, US Patent 5883177, Mar 16, 1999.
9. Brothers P. D., Morgan R. A., Amorphous fluoropolymer containing perfluoro(ethyl vinyl ether, EP 0925314B1, 11.09.1997.
10. Triulzi F., Arcella V., Tortelli V., Calini P., Amorphous perfluorinated copolymers, EP 1256592A1, 13.11.2002.
11. Arcella V., Apostolo M., Triulzi F., Amorphous perfluorinated copolymers, EP 1469015A1, 08.04.2004.
12. Apostolo M., Triulzi F., Arcella V., Tortelli V., Calini P., Amorphous perfluorinated copolymers, US Patent 6936668B2, Aug. 30, 2005.
13 Sokolov V. I., Boyko V. E., Goryachuk I. O., Igumnov S. M., Molchanova S. I., Pogodina Yu. E., Polunin E.V., Synthesis and optical properties of copolymers of perfluoro-2,2-dimethyl-1,3-dioxole and perfluoropropyl vinyl ether, Russian Chemical Bulletin, International Edition, 2017, 66(7), 1284-1289.
14. Polunin E. V., Molchanova S. I., Pogodina J. E., Sokolov V. I., Zavarzin I. V., Homo- and co-polymerisation of perfluoroisopropylvinyl ether under high pressure, Fluorine Notes, 2017, 114, 5-6.
15. Sokolov V. I., Goriachuk I. O., Zavarzin I. V., Molchanova S. I., Pogodina Yu. E., Polunin E. V, Yarosch A. A., New copolymers of perfluoro-2-ethyl-2-methyl-1,3-dioxole and perfluorovinyl ether with low non-monotonic refractive index, Russian Chemical Bulletin, International Edition, 2019, 68(3), 559-564.
ARTICLE INFO
Received 07 September 2022
Accepted 23 September 2022
Available online October 2022
Recommended for publication by PhD V. L. Don
eLIBRARY Document Number (EDN) ZYZLQI

Fluorine Notes, 2022, 144, 3-4
