Received: June 2020
DOI 10.17677/fn20714807.2020.04.03
Fluorine Notes, 2020, 131, 5-6
ACTIVATION ENERGY AND THERMAL EFFECT OF INITIATION REACTION CATIONIC POLYMERIZATION OF p-METHYLSTYRENE IN THE PRESENCE OF COMPLEX CATALYSTS: BORON FLUORIDE - HYDROGEN FLUORIDE AND BORON FLUORIDE - WATER IN TOLUENE
1Babkin V.A., 1Andreev D.S., 1Ignatov A.V., 2Belousova V.S., 3.4 E.S. Titova., 3,4Titova E.S., 3Rakhimov A.I., 3Rakhimova N.A., 5G.E. Zaikov
1Volgograd State Technical University (Sebryakovsky br.), 403343 Volgograd Region, Mikhailovka, Michurina st., 21
e-mail: babkin_v.a@mail.ru
2I.M. Sechenov 1st Moscow State Medical University, 119991, st. Trubetskaya, 8, bld. 2
3Volgograd State Technical University, 400005 Volgograd, Lenin av., 28
e-mail: organic@vstu.ru
4Volgograd State Medical University, 400131 Pavshikh Bortsov sq., 1
5N. M. Emanuel Institute of Biochemical Physics of RAS, 119334, Moscow, Kosygina st., 4,
e-mail: chembio@sky.chph.ras.ru
Abstract: This paper presents data on activation energy and thermal effects at the initiation stage of cationic polymerization of p -methylstyrene in the presence of complex catalysts: boron fluoride - hydrogen fluoride and boron fluoride – water in toluene having various stoichiometric composition. We studied the systems with following stoichiometric compositions BF3-HF (H2O) - p‑methylstyrene-toluene, respectively: 1: 1: 1 (in the absence of toluene), 1: 1: 1: 1; 1: 1: 1: 2; 1: 1: 1: 3 and 1: 1: 1: 4, by quantum chemical calculation using the ab ininto method. It was shown that for BF3-HF- p-methylstyrene - toluene system (I), the studied stoichiometric composition does not affect the activation energy of initiation. For BF3-H2O - p-methylstyrene - toluene system (II) system was established, that with toluene amount increase in this system the activation energy decreases.
Keywords: p-methylstyrene, protonation, boron fluoride - hydrogen fluoride catalyst, thermal effect of reaction, ab initio method.
Introduction
It is well known that toluene is an indifferent solvent widely used in various chemical reactions and, in particular, in the cationic polymerization reaction of various olefins [1]. Within traditional meaning the indifferent solvents are solvents whose interaction energy of molecules with a dissolved substance is no less than an order of magnitude lower than the solvation energy [2]. In this regard, often, especially when studying (modeling) the mechanisms of various chemical reactions, the influence of indifferent solvents was practically not taken into account and equated to the same reactions, but which take place in vacuum or in the absence of a solvent (for example see [3]). Nevertheless, it has already been noted that in indifferent solvent the reaction rates often differ from the rates that occur in the absence of solvents [4].
In this regard, the following tasks were set: 1) to ferret out the influence of indifferent solvent on mechanism and energetics of reaction, 2) to determine the thermal effects and activation barriers in initiation reaction of cationic polymerization of p-methylstyrene in the presence of complex catalysts BF3-HF and BF3-H2O in toluene depending on amount of toluene. For this, we studied the systems BF3-HF- p-methylstyrene-toluene (I) and BF3-H2O-p-methylstyrene-toluene (II) with following stoichiometric composition - 1(BF3):1 (HF or H2O): 1(p-methylstyrene):1 (toluene), 1:1:1:2, 1:1:1:3, 1:1:1:4.
It is advisable to theoretically estimate the values of activation barriers and thermal effects by quantum chemical calculation of studied initiation mechanisms of cationic polymerization for p‑methylstyrene due to the fact that experimental methods can be very difficult (and often impossible) to determine the activation energy
Methodical part
To study the initiation mechanism of cationic polymerization for p-methylstyrene in the presence of above complex catalysts, the classical quantum-chemical ab initio method in the 6‑311G** basis [5-6] was chosen as one of the best methods for estimating the activation energies and thermal effects of studied reactions [7-8]. When modeling of processes, the MacMolPlt program [9] was also used.
The initiation mechanism was studied in one coordinate (similarly to [10-12]). As the reaction coordinate for models (I) and (II) the RC(1)–H(20) bond was chosen. Multiplicity M = 1, because M = 2S + 1, where S is the resultant spin equal to 0 due to the fact that all electrons are paired. Throughout the interaction path of catalyst and p-methylstyrene and at each stage, the charge conservation law was fulfilled, i.e. the distributed total charge on all atoms is zero. The simulation was carried out in accordance with Markovnikov rule, i.e. the proton attacks the most hydrogenated carbon atom of p-methylstyrene - C(1).
Calculation data
The initial models (I) and (II) with stoichiometric composition 1:1:1:1 are presented in Fig. 1 and 5, and final structures (after the interaction of catalyst with a monomer) are shown in Fig. 2 and 6. Changes in total system energy along the reaction coordinate R are shown in Fig. 3 and Fig.7, and changes in charges at the atoms - in Fig. 4 and Fig 8. The values of activation energies and thermal effects for different stoichiometric composition of components are presented in Table 1 and Table 2.
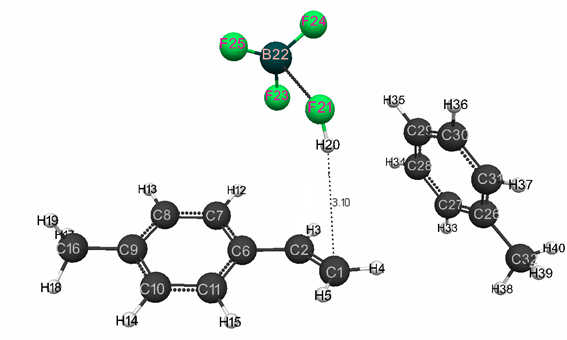
Figure 1. Structure of original model (I) of complex catalyst HF·BF3 with p-methylstyrene in toluene having stoichiometric composition 1:1:1:1.
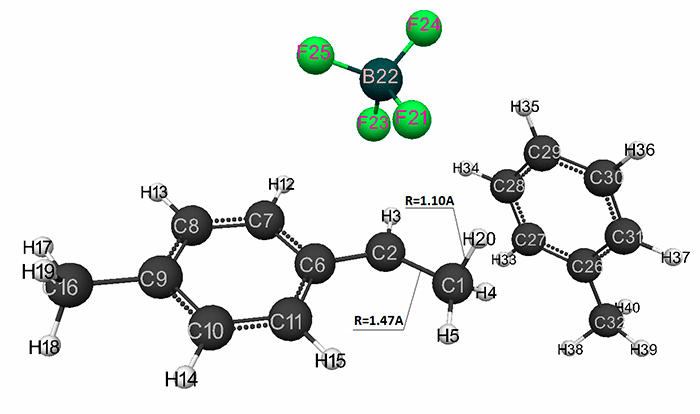
Figure 2. Final structure of complex catalyst HF·BF3 interaction with p-methylstyrene in toluene having stoichiometric composition 1:1:1:1.
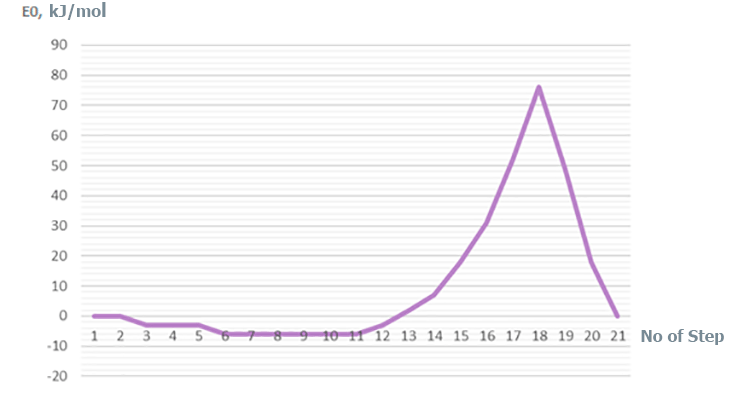
Figure 3. Change in total energy (E0) of studied reaction (1st -21th stages of interaction).
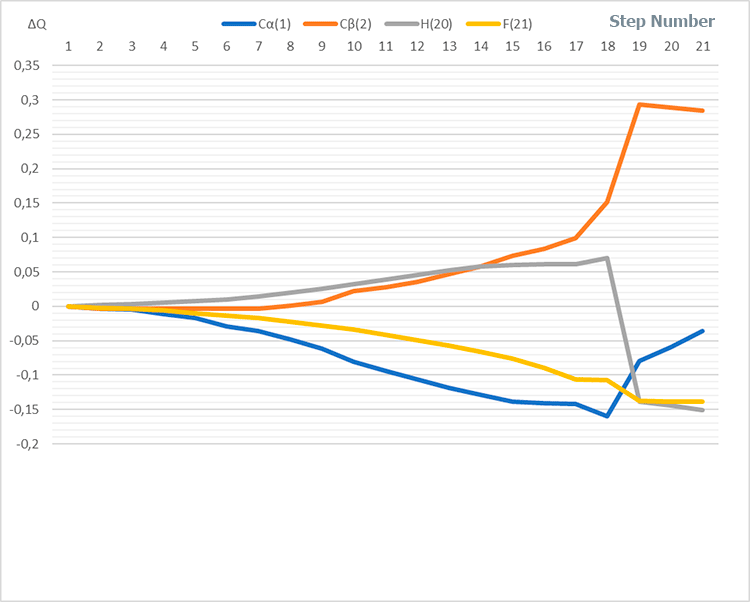
Figure 4. Change of charges along reaction coordinate in system (I) having stoichiometric composition 1:1:1:1.
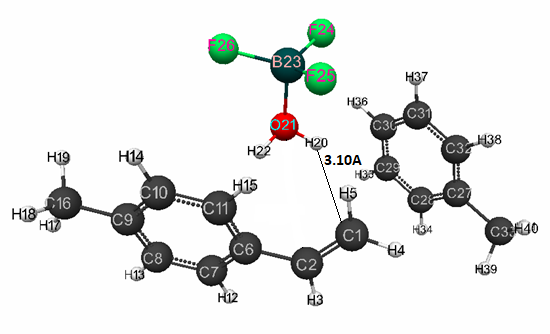
Figure 5. Initial model structure of complex catalyst H2O·BF3 with p-methylstyrene in toluene having stoichiometric composition 1:1:1:1.
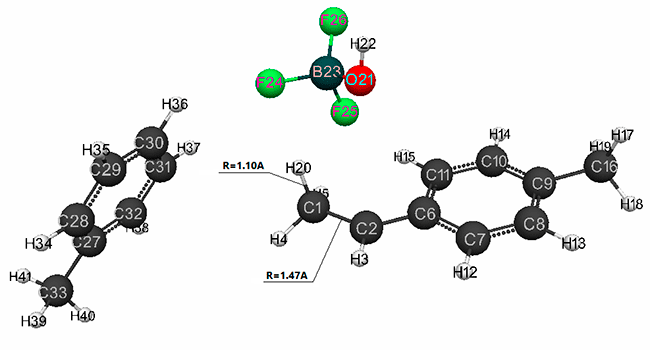
Figure 6. Final interaction structure of complex catalyst H2O·BF3 with p-methylstyrene in toluene having stoichiometric composition 1:1:1:1.
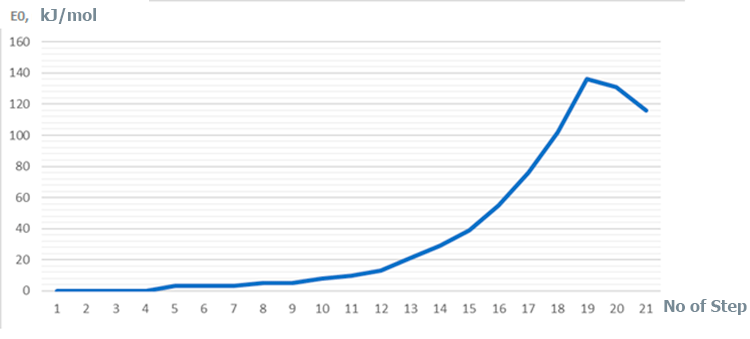
Figure 7. Change in total energy (E0) of studied reaction (1st -21th-stages of interaction).
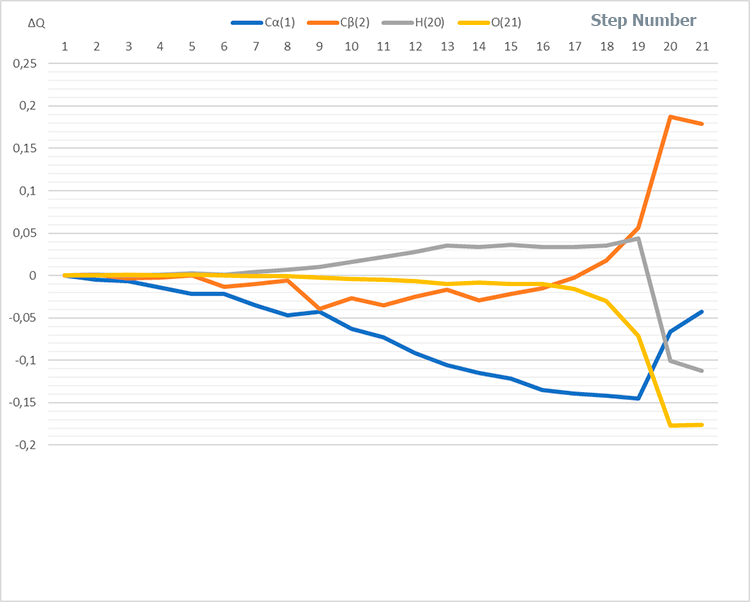
Figure 8. Change in charges along reaction coordinate in system (II) having stoichiometric composition 1:1:1:1.
Based on analysis of catalyst and monomer fragments behavior - breaking of a H-F bonds, formation of new ones (H-Cα), changes in total energy of molecular system along reaction coordinate (see Fig. 3) and charge redistribution (see Fig. 4) - it can be seen that in the former case (for BF3-HF system, i.e. for p-methylstyrene–toluene) the initiation mechanism is the usual acceptance of a proton from the BF3-HF complex, and its attachment to the most hydrogenated atom of p-methylstyrene, with simultaneous transformation of π-double bond of monomer to a single σ-bond (similarly to [10]).
In this case, as follows from Table 1, the amount of toluene practically does not affect the activation energy (it varies within the range of methodic error in the range of 73-79 kJ/mol), and also does not affect the initiation mechanism of cationic polymerization of p-methylstyrene in the presence of complex catalyst BF3-HF.
Table 1. Activation energy (Еа) and thermal effects (Q) of reaction in system (I).
|
No |
Stoichiometric composition of molecular system BF3-HF - p-methylstyrene |
Еа, kJ/mol |
Q, kJ/mol |
|
1 |
1:1:1:0 |
79 |
8 |
|
2 |
1:1:1:1 |
76 |
0 |
|
3 |
1:1:1:2 |
73 |
0 |
|
4 |
1:1:1:3 |
73 |
-6 |
|
5 |
1:1:1:4 |
78 |
-3 |
It should be noted the following trend: an increase in the amount of solvent – i.e. toluene, can change the nature of reaction - from endothermic to exothermic.
Table 2. Activation energy (Еа) and thermal effects (Q) of reaction in system (II).
|
№ |
Stoichiometric composition of molecular system BF3- H2O - p-methylstyrene |
Еа, kJ/mol |
Q, kJ/mol |
|
1 |
1:1:1:0 |
134 |
115 |
|
2 |
1:1:1:1 |
136 |
116 |
|
3 |
1:1:1:2 |
123 |
86 |
|
4 |
1:1:1:3 |
116 |
76 |
|
5 |
1:1:1:4 |
118 |
86 |
Based on analysis of complex catalyst BF3-H2O and monomer fragments behavior - breaking of a O-H bond, transformation of π-bond of monomer into a σ-bond, and also formation of new О-С and С-H bonds, changes in total energy of molecular system along reaction coordinate (see Fig. 4), change in charges during reaction (see Fig. 6) - it can be seen that in this case the initiation mechanism is the coordinated process, accompanying by simultaneous breaking and formation of above bonds (similarly to [12]). In this case, the amount of toluene practically does not affect the initiation mechanism, but it very significantly affects the energy of this reaction. Regardless of the amount of toluene, the reactions are endothermic and have a barrier character. In this regard, the following dependence has been established: an increase in the amount of toluene in reaction system by 4 times reduces the activation energy from 134 kJ/mol to 116 kJ/mol, and the thermal effect of reaction - from 116 kJ/mol to 76 kJ/mol, respectively.
Obviously, the obtained results of quantum-chemical calculations at the nanoscale level for both systems (I and II) should be experimentally verified.
Conclusion
Thus, we first performed systematic studies of cationic polymerization initiation mechanism of p-methylstyrene in the presence of complex catalysts (BF3-HF (I) and BF3-H2O (II) in toluene) in systems having different stoichiometric composition. It was found that for system (I), the amount of toluene does not affect the activation energy of this reaction, and the thermal effect increases with an increase in the amount of toluene; in such a case, this reaction can even turn from exothermic to exothermic.
In system (II), an increase in the amount of toluene leads to a decrease in activation energy and thermal effect.
Ultimately, the obtained dependences (together with the well-known method of controlling the olefin initiation reaction by varying the nature of ligand environment of Lewis acid (BF3) and Bronsted (HF, H2O) [13]) give new possibilities for controlling of reaction under study by changing the ratio between the catalyst and solvent (toluene) and also to obtain the polymer (p-methylstyrene) with predetermined properties.
References
- Kennedy, J., Cationic polymerization of olefins. - Publishing house "Mir": M., 1978, 431 p.
- Handbook of a chemist, 21. Chemistry and chemical technology. https://www.chem21.info/info/1490027/.
- Matsevich O. V., Accounting for solvent in quantum-chemical modeling of synthesis mechanism of polyarylenephthalides, Bulletin of Kazan Technological University, 2014, 17 (10), 24-26.
- Babkin V. A. et al., Quantum-chemical aspects of cationic polymerization of olefins, 1996, Gilem Publishing House (Ufa), 188 pp.
- Schmidt M. W. et al., General Atomic and Molecular Electronic Structure System, J. Comput. Chem. 1993, 14, 1347-1363.
- Granovsky A. A., Firefly version, v. 8, 2013. http://classic.chem.msu.su/gran/firefly/index.html
- Ermakov A. T., Quantum mechanics and quantum chemistry, Yurayt Publishing House, 2016, 555 pp.
- Tsirelson V. G., Quantum chemistry: molecules, molecular systems and solids, Binom Publishing House, 2010, 422 pp.
- Bode B. M., Gordon M. S., MacMolPlt, A graphical user interface for GAMESS, Journal of Molecular Graphics, 1998, 16, 133-138.
- Babkin V. А. et al., Study of the interaction of the complex catalyst HF-BF3 with p‑methylstyrene by AB INITIO, Fluoride Notes, 2020, 128, 1-2.
- Babkin V. А. et al., Study of interaction mechanism of complex catalyst aluminum chloride - hydrochloric acid and p-methylstyrene in toluene, Bulletin of Kazan Technological University, 2020, 23 (1), 9-12.
- Babkin V. А. et al., Quantum chemical calculation of cationic polymerisation initiation mechanism of propylene with chloride, Aluminum Aquacomplex, Oxidation Communications, 2020, 43 (1), 24-29.
ARTICLE INFO
Received 17 June 2020
Accepted 24 June 2020
Available online August 2020
Recommended for publication by PhD M. A. Manaenkova
Fluorine Notes, 2020, 131, 5-6
