Received: December 2017
DOI 10.17677/fn20714807.2018.01.04
Fluorine Notes, 2018, 116, 7-8
Polymerization of α-fluoroacrylates and fabrication of surface relief structures by UV exposure without photoinitiators
Viktor I. Sokolova,*, Nikita M. Bityurinb, Svetlana I. Molchanovaa, Alexander V. Pikulinb, Andrey A. Tyutyunovc, Sergey M. Igumnovc
a Federal Scientific Research Centre “Crystallography and Photonics”, Russian Academy of Sciences, Leninsky ave. 59, 119333 Moscow, Russia
b Institute of Applied Physics, Russian Academy of Sciences, Ul'yanov St. 46, 603950 Nizhny Novgorod, Russia
c Institute of Organoelement Compounds, Russian Academy of Sciences, Vavilova St. 28, 119991, GSP-1, Moscow, V-334, Russia
*corresponding author: visokol@rambler.ru
Abstract: New -fluoroacrylic monomers with a fluorination degree of 83.3 – 90.9% have been recently synthesized. It was found that the monomers have high reactivity in the radical polymerization process, which can be launched without adding special photoinitiators by exposure to actinic radiation of wavelength λ ≤ 270 nm. In the process of photopolymerization, the penetration depth of UV light into -fluoroacrylates was shown to increase, indicating strong bleaching during the irradiation. The monomers and corresponding homopolymers exhibit high optical transparency in the 840, 1310 and 1550 nm telecom wavelength regions. Surface relief structures were fabricated by UV exposure of α-fluoroacrylates through the photomask without any photoinitiator.
Keywords: fluorinated polymer, photopolymerization, photobleaching, UV lithography, telecommunications, integrated optics
1. Introduction
Fluorinated polymers have become important for the development of various integrated optical elements due to their high optical transparency in all three telecom wavelength regions around 840, 1310 and 1550 nm [1-4]. For example, by using fluorinated polymers, the optical waveguides with propagation loss 0.07 dB/cm at λ=1550 nm have been demonstrated [5]. Theoretical calculations reveal that amorphous perfluorinated polymers can have an even lower absorption coefficient of 0.15 dB/km in this wavelength range [6]. The high transparency of fluoropolymers in the visible and near IR spectral regions is due to the fact that vibrational overtones of C-F bonds are shifted to longer wavelengths as compared with those of C-H bonds [2]. As a result, fluorinated polymers show negligible absorption loss in the telecom spectral regions [7]. Additionally, α-fluoroacrylates exhibit higher shock resistance, as well as moisture and thermal stability [8], since the energy of CF bond is greater than that of СН bond.
The polymer relief structures (channel waveguides, grippers, grooves, micro lenses, etc.) are usually fabricated by means of the radical photopolymerization of liquid monomers or viscous mixtures of monomers and oligomers (prepolymers) upon UV exposure by adding special photoinitiators (PI) to the formulation. The PI molecule absorbs the incident photon and generates reactive species, which launch the radical polymerization process. However, the photoinitiator can lead to yellowing and diminishing of the optical transparency of the polymer. Besides, many photoinitiators are poorly dissolved in highly fluorinated monomers. On the other hand, both the experiment [9 - 11] and quantum chemical modeling [12] have shown that the excitation of methacrylate and acrylate molecules by high-energy photons can generate reactive radicals resulting in the self-initiation of photopolymerization reactions. This opens up new possibilities for the fabrication of polymer surface structures.
In the present paper, we demonstrate the possibility of the UV ( λ ≤270 nm) photopolymerization of formulations that contain the highly fluorinated monomers and do not contain PIs. The effect of bleaching of the formulation for the UV actinic light during the polymerization is studied. To our knowledge, a similar phenomenon has been observed earlier only for acrylate-based hydrocarbon resins [10, 11]. The photobleaching effect is caused by the conversion of the C=C part of C=C-C=O conjugated system during the polymerization process.
Surface structures with width 50 μm and height 25 μm were fabricated by UV exposure without photoinitiator using α-fluoroacrylate prepolymers.
2. Materials
To provide the maximal optical transparency of polymers in the telecom spectral regions, especially in the С – band (1530 – 1565 nm), one should increase the degree of fluorination of the monomers. On the other hand, the monomers must have high reactivity in the process of radical polymerization, which permits to fabricate surface relief structures using UV photolithography or direct laser writing. To meet these requirements, we synthesized new -fluoroacrylate monomers with fluorination degree 83.3 – 90.9 % based on tertiary alcohols. The monomers of the homologous series
CH2=CF-COO-C(CF3)2-(CF2)m-CF3, (1)
where m = 0, 3, 5 were synthesized by using the method described in Refs. [7-8]. The monomers (1) are colorless transparent liquids with low viscosity. Some physical properties of these monomers and corresponding homopolymers are given in Table 1.
Table 1. Physical properties of fluorinated monomers (1) and corresponding homopolymers. nD is refractive index at λ= 589.3 nm, measured at 20°C.
|
Monomer CH2=CF-COO-C(CF3)2-(CF2)m-CF3 |
m = 0 |
m = 3 |
m = 5 |
|
Degree of fluorination, % |
83.3 |
88.9 |
90.9 |
|
nD (monomer) |
1.3159 |
1.3136 |
1.3127 |
|
nD (polymer) |
1.3475 |
1.3451 |
1.3438 |
|
Boiling temperature, 0С (mercury pressure, mm) |
5 (10) |
75 (10) |
20 (1) |
|
Shore hardness number, D scale (polymer) |
83 ± 2 |
79 ± 2 |
77 ± 2 |
Typical absorption spectra of α-fluoroacrylic monomers and homopolymers in the telecom wavelength regions are shown in Fig. 1. One can see that the absorption coefficient equals 0.02, 0.04 and 0.1 dB/cm at λ= 840, 1310 and 1550 nm, respectively. The low absorption of α-fluoroacrylates (1) is due to the high fluorination degree, which exceeds 83%.
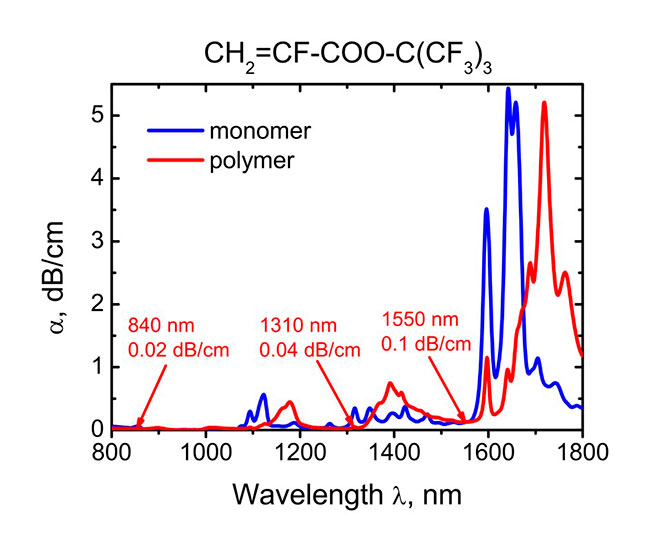
Fig. 1. Absorption coefficient α(λ) of the monomer CH2=CF-COO-C(CF3)3 (1) and corresponding homopolymer (2) in the 840, 1310 and 1550 nm telecom wavelength regions.
3. Experiment
3.1. Self-initiation of the radical photopolymerization of -fluoroacrylates
Polymer surface relief structures can be fabricated on various substrates by polymerizing acrylate monomers or viscous prepolymers using UV photolithography or direct laser writing [13-15]. To start radical photopolymerization reaction, the generation of primary radicals is necessary. This requires an addition of the specific photoinitiator to the formulation at a typical concentration of 0.5 – 1 wt %. However, the addition of the photoinitiator is not desirable, since it may result in yellowing and diminishing of optical transparency of the polymer. On the other hand, it is known that illumination of hydrocarbon methacrylate and acrylate molecules with high-energy photons can generate •C-C• biradicals resulting in the self-initiation of radical photopolymerization reaction [9 - 12].
To investigate whether the polymerization reaction of highly fluorinated α-fluoroacrylates can be self-initiated upon UV exposure and to determine the wavelength below which the actinic radiation can launch the self-initiation process, we irradiated monomers (1) with various UV light sources. These included an Nd-YAG (263 nm) laser, as well as a high-pressure 150 W xenon lamp equipped with bandpass interference filters. It was found out experimentally that polymerization of α-fluoroacrylates can be self-initiated by UV light with a wavelength below 270 nm. This is close to λ= 267 nm calculated for acrylates using quantum chemical modeling [12]. The possible mechanism of the self-initiation of radical polymerization reactions is direct excitation of the conjugated С=С-C=O system of α-fluoroacrylates by the short-wavelength radiation, which results in the formation of reactive •C-C• biradicals. The UV absorption spectra of the monomers show (see Fig. 2) a wide band centered at 207 nm, which can be attributed to the electronic transitions in the conjugated С=С-C=O system [12]. The spectra of the monomer solutions in hexane were measured with respect to the pure solvent in a 1-cm thick quartz cuvette.
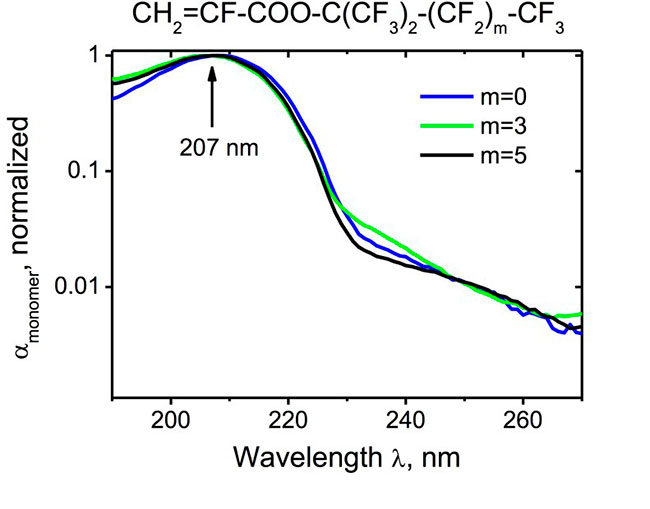
Fig. 2. Normalized absorption coefficients αmonomer of α-fluoroacrylic monomers CH2=CF-COO-C(CF3)2-(CF2)m-CF3 with m = 0, 3, 5 in the UV spectral range.
In the process of photopolymerization, which is accompanied by the conversion of С=С double bonds and formation of polymer macromolecules, the bleaching of the formulation can take place. Earlier, the bleaching has been observed experimentally only for methacrylates and acrylates [10, 11] and theoretically described in Ref. [16]. Here, we explored for the first time this process for α--fluoroacrylates.
Monomers (1) without photoinitiator were illuminated by the low-intensity actinic radiation of wavelength 250 - 270 nm in a 1-mm thick quartz cuvette. Figure 3a shows the transmission T(λ) of UV light through the cuvette with CH2=CF-COO-C(CF3)2-(CF2)3-CF3 monomer, and Figure 3b shows the refractive index nD of this monomer versus exposure time t. One can see that during the irradiation, the curve T(λ) is being shifted to the shorter wavelengths, clearly indicating the bleaching of the formulation. Moreover, its refractive index increases monotonically. Similar results were also obtained for other monomers of the homologous series (1). It follows from Fig. 3 that the kinetics of the radical photopolymerization of α-fluoroacrylates is characterized by the induction period due to the quenching of the free radicals by inhibitors (probably by oxygen dissolved in the monomer). The induction period is followed by an accelerated conversion step which is finally saturated.
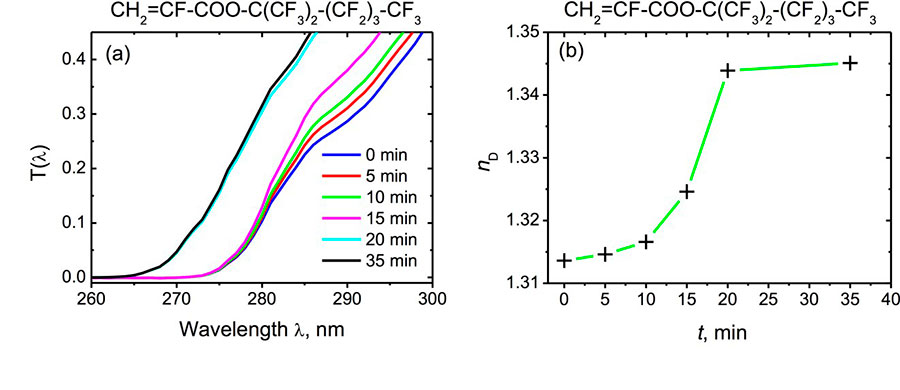
Fig. 3. Transmission coefficients T(λ) of 1-mm long quartz cuvette with monomer CH2=CF-COO-C(CF3)2-(CF2)3-CF3 (a) and refractive index nD of the monomer (b) as a function of exposure time.
We attribute the increase of the refractive index to the shrinkage of the monomer, and the bleaching to the conversion of the acrylic C = C double bonds during the radical photopolymerization process. Indeed, Figure 4 demonstrates the effect of the UV exposure on the С=С-C=O absorption band centered at 207 nm. The transmission spectra of the monomer CH2=CF-COO-C(CF3)3 shown in Fig. 4 were recorded in a specially designed micro-cuvette of thickness 10 m with a Cary-50 spectrophotometer. One can see that the absorption band strongly decreases with time t , clearly indicating a decrease in the concentration of C = C double bonds during UV exposure.
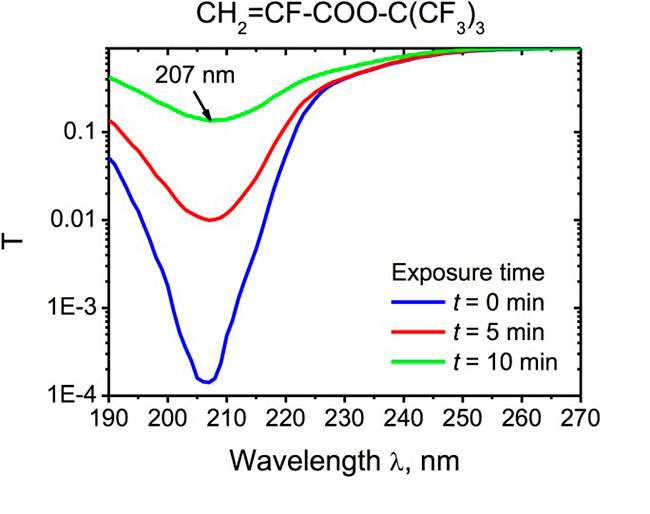
Fig. 4. Transmission coefficient T(λ) of 10-m thick quartz micro-cuvette with monomer CH2=CF-COO-C(CF3)3 in the process of UV exposure.
3.2. Preparation of viscous α-fluoroacrylate prepolymers
The formation of polymer surface structures with UV actinic radiation is more effective if the monomer has a sufficiently high viscosity. The α-fluoroacrylates (1) are characterized by low viscosity, therefore it should be increased before use by preparation of a prepolymer. This can be done upon UV exposure without any photoinitiator. The viscosity of the prepolymers can be controlled directly with a viscosimeter, or through measuring refractive index of the prepolymer, see Fig. 3b above. One should note that homopolymers fabricated with monomers (1) are readily dissolved in these monomers. This permits to fabricate the formulations with required viscosity by mixing monomer and homopolymer in certain weight ratios.
3.3. Fabrication of polymer surface structures by UV exposure of -fluoroacrylates
To fabricate surface relief structures, we used UV-curable prepolymers (1). No conventional photoinitiator was added to the formulation. At first, the polymer buffer layer was fabricated on a silicon substrate. A small amount of the formulation was placed on the surface of buffer layer and covered with a photomask (quartz plate with 100-nm chromium layer). Then, the formulation was irradiated by UV light (see Fig. 5). After curing, the structure was washed in an organic solvent to remove the non-polymerized formulation.
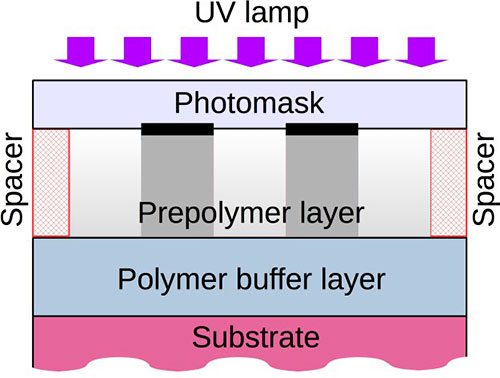
Fig. 5. Scheme of relief structure fabrication by UV exposure of the viscous prepolymer through the photomask.
Figure 6 demonstrates a photograph of the polymer relief structure fabricated by UV curing of α-fluoroacrylate monomer CH2=CF-COO-C(CF3)3 without photoinitiator. The width of the rectangular bars and the distance between them equals 50 μm, the height of the bars is μ25 m. Relief structures with such dimensions can be used as multimode light-guiding cores, grippers or grooves for fabricating polymer inverted rib waveguides.

Fig. 6. Photograph of the polymer relief structure, fabricated by UV exposure of α-fluoroacrylate CH2=CF-COO-C(CF3)3 through the photomask without photoinitiator. Top view.
4. Discussion
The selection of the optimal wavelength of actinic radiation is very important in the process of polymer structures formation. If the depth of penetration of UV light into prepolymer is less than the height of the structure, the relief structure cannot be formed. Alternatively, if the penetration depth is much greater than the height of the structure, only a small part of actinic radiation is involved in the curing process. Another part, due to the scattering and reflection from the buffer layer, substrate, etc. can get into the dark areas under the photomask and initiate the polymerization process there. This leads to the deterioration of the resolving power of the lithographic process. For example, the penetration depth of 260 nm light into CH2=CF-COO-C(CF3)3 monomer equals 18 μm, whereas that into the corresponding homopolymer is 140 μm. Therefore, one can conclude that the UV light in the spectral range between 250 and 270 nm is optimal for fabricating polymer surface structures with the height 25 - 100 μm. To form relief structures with lower heights, one should use shorter wavelength radiation, which has a smaller penetration depth, or suppress the undesired polymerization in the dark areas by different means, e.g., by introducing the diffusing radical quencher to the composition [17].
5. Conclusions
This study reports on investigations of new UV curable -fluoroacrylic monomers with fluorination degree 83.3 – 90.9 %, possessing high optical transparency in the telecom wavelength regions. It was shown that the radical polymerization of -fluoroacrylates can be self-initiated by exposure to UV light of a wavelength shorter than 270 nm. The photopolymerization is accompanied by strong bleaching in the UV range, which is due to conversion of acrylate С=С double bonds. By using UV photolithography, we have successfully fabricated polymer surface structures without any special photoinitiator. The method can be used for fabricating various integrated optical elements: light-guiding cores, grippers, grooves, etc.
Acknowledgements
This work was supported by Russian Science Foundation (RSF) grant 14-19-01659.
References
- M. Zhou, Low-loss polymeric materials for passive waveguide components in fiber optical telecommunication, Opt. Eng. 41 (2002) 1631-1643.
- W. Groh, Overtone absorption in macromolecules for polymer optical fibers, Makromol. Chem. 189 (1988) 2861–2874.
- H. Ma, A.K.-Y. Jen, L.R. Dalton, Polymer-based optical waveguides: materials, processing, and devices, Adv. Mater. 14 (2002) 1339–1365.
- G. Li, J. Wang, S. Guixian, X. Jian, L. Wang, M. Zhao, Synthesis and characterization of fluorinated crosslinkable poly(phthalazinone ether)s bearing tetrafluorostyrene groups for optical waveguides, Polymer Journal 42 (2010) 880–886.
- A. Yeniay, R. Gao, K. Takayama, R. Gao, A. F. Garito, Ultra-low-loss polymer waveguides, J. Lightwave Technol. 22 (2004) 154– 158.
- W. Groh, A. Zimmermann, What is the lowest refractive index of an organic polymer? Macromolecules 24 (1991) 6660–6663.
- A.A. Tyutyunov, V.E. Boyko, A.V. Sin’ko, S.M. Igumnov, S.I. Molchanova, E.V. Khaidukov, V.I. Sokolov, Synthesis of Perhalogenated tert-Butyl-α-fluoroacrylates and their Optic Characteristic Data, Fluorine Notes 6 (2014). http://notes.fluorine1.ru/public/2014/6_2014/letters/letter3.html.
- L.S. Boguslavskaya, I.Yu. Panteleeva, T.V. Morozova, A.V. Kartashov, N.N. Chuvatkin, α-fluoroacrylates: synthesis, properties and use, Rus. Chem. Rev. 59 (1990) 906–917.
- T. Scherzer, Photopolymerization of acrylates without photoinitiators with short-wavelength UV radiation: A study with real-time fourier transform infrared spectroscopy, J. Polym. Sci. Part A Polym. Chem. 42 (2004) 894–901. doi:10.1002/pola.11039.
- T. Scherzer, VUV-induced photopolymerization of acrylates, Macromol. Chem. Phys. 213 (2012) 324–334. doi:10.1002/macp.201100485.
- A.P. Alexandrov, S.V. Muraviov, N.A. Babina, N.M. Bityurin, Formation of 3D dielectric structures by initiating polymerization with the fourth harmonic of an Nd laser, in: M.N. Libenson (Ed.), Proc. SPIE 10th Int. Conf. Nonresonance Laser-Matter Interact., St.Petersburg, Russia, 2001: pp. 74–78. doi:10.1117/12.431205.
- F. Bauer, U. Decker, S. Naumov, C. Riedel, Photoinitiator-free UV curing and matting of acrylate-based nanocomposite coatings: Part 3, Prog. Org. Coatings. 77 (2014) 1085–1094. doi:10.1016/j.porgcoat.2014.03.013.
- L. Eldada, L.W. Shacklette, Advances in polymer integrated optics, IEEE J. Sel. Top. Quantum Electron. 6 (2000) 54–68. doi:10.1109/2944.826873.
- M.-C. Oh, K.-J. Kim, W.-S. Chu, J.-W. Kim, J.-K. Seo, Y.-O. Noh, H.-J. Lee, Integrated photonic devices incorporating low-loss fluorinated polymer materials, Polymers 3 (2011) 975-997.
- S.M. Igumnov, V. I. Sokolov, V.K. Men’shikov, O.A. Mel’nik, V.E. Boiko, V.I. Dyachenko, L.N. Nikitin, E.V. Khaidukov, G.Yu. Yurkov, V.M. Buznik, Fluorinated monomers and polymers with specific properties for integrated optics and photonics, Doklady Chemistry 446 (2012) 183-187.
- N. Bityurin, A. Pikulin, A. Alexandrov, Modeling of bleaching wave regime of UV laser polymerization of acrylates without initiators, Appl. Surf. Sci. 208–209 (2003) 481–485. doi:10.1016/S0169-4332(02)01438-1.
- A. Pikulin, N. Bityurin, V.I. Sokolov, Model of diffusion-assisted direct laser writing by means of nanopolymerization in the presence of radical quencher, AIP Adv. 5 (2015) 127215. doi:10.1063/1.4938512.
Recommended for publication by PhD Andrey Tyutyunov
Fluorine Notes, 2018, 116, 7-8
