Received: June 2017
DOI 10.17677/fn20714807.2017.03.03
Fluorine Notes, 2017, 112, 5-6
Surprising chloropentanfluorobenzene preparation from pentafluorobenzene, chlorosulfuric acid and iodine
V.E. Boikoab, A.V. Edunovb, S.М. Igumnovab
aA.N. Nesmeyanov Institute of Organoelement Compounds, Russian Academy of Sciences, Russia, 119991, GSP-1, Moscow, V-334, Vavilova St, 28
b SIA “P&M-Invest” ltd, Russia, 119334 Moscow, Leninsky prospect 47
e-mail: boykii@mail.ru
Abstract: Chloropentafluorobenzene is obtained trough interaction between pentafluorobenzene and chlorosulfuric acid in the presence of iodine or iodine chloride. The interaction between pentafluorobenzene and chlorosulfuric acid and iodine bromide results in obtaining of bromopentafluorobenzene.
Key words: halogenopolyfluorobenzenes, chloropentafluorobenzene, bromopentafluorobenzene, chlorosulfuric acid
Halogenopolyfluorobenzenes, in particular iodopentafluorobenzene, are starting compounds for the obtaining of fluoroaromatic compounds with different functional groups. Compounds, containing pentafluorophenyl fragment, are used in medicinal chemistry, electrotransport devises, light-emitting diodes, polygraphy, LCD displays, fluorine two-phase ligands [1,2].
Iodopentafluorobenzene is obtained by the iodation of pentafluorobenzene in 65 % [3] or in 20 % oleum with 71 % yield [4], by the interaction between pentafluorophenylmagnesium chloride and elemental iodine [5] , by the reaction of pentafluorophenyllithium or potassium with iodine in demethylformamide with 85 % yield [6] , by the interaction between pentafluorobenzene and triiodoisocyanuric acid in 98 % sulfuric acid with yield 74 % [7].
We assumed to obtain iodopentafluorobenzene by the interaction between pentafluorobenzene and iodine chloride in the presence of chlorosulfuric acid.
However, we found out, that pentafluorobenzene reacts with chlorosulfuric acid in the presence of iodine chloride or elemental iodine within the temperature range from 700C to 90 0C with the formation of mixture of chloropentafluorobenzene, iodopentafluorobenzene and 1,2-dichloropentafluorobenzene (Scheme 1), where chloropentafluorobenzene is the main product.
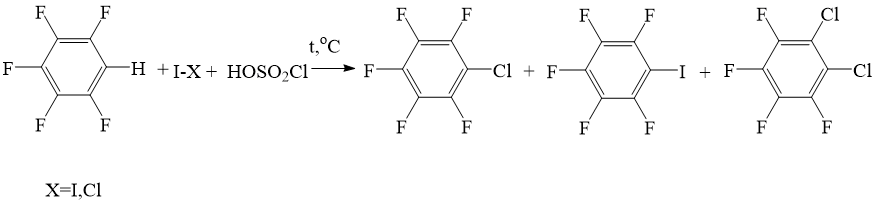
Scheme 1
Further when attempting to increase the yield of iodopentafluorobenzene, it was studied the influence of temperature and ratio of reagents and additives per proportion of reagents. The obtained data of reaction products after 2 hours by different proportion of reagents are summarized in Table 1.
Table 1. Influence of ratio of reagents and temperature on the composition of reaction products.
|
# Experiment |
Ratio of regents (relative to PhfH) |
ToC |
Ratio of compound in final mixture |
|||||
|
I-X /mol |
Mol HOSO2Cl |
Mol SOCl2 |
||||||
|
PhfCl |
PhfCl2 |
PhfI |
PhfH |
|||||
|
1 |
ICl / 1.2 |
3 |
– |
70 |
73 |
6.8 |
10.2 |
– |
|
2 |
ICl / 1 |
2.5 |
– |
90 |
73.5 |
6.9 |
19.6 |
– |
|
3 |
I2 / 1.2 |
3 |
– |
64 |
80 |
10 |
10 |
– |
|
4 |
I2 / 0.6 |
3 |
– |
60 |
68.3 |
14.6 |
17.1 |
– |
|
5 |
I2 / 0.15 |
3 |
– |
90 |
60.9 |
13.8 |
25.3 |
– |
|
6 |
I2 / 0.1 |
3 |
– |
90 |
44.8 |
6.2 |
14.6 |
34.4 |
|
7 |
I2 / 0.05 |
3 |
– |
70 |
7.2 |
– |
– |
92.8 |
|
8 |
I2 / 0.25 |
1.5 |
1,5 |
70 |
24 |
3.3 |
6.6 |
64.1 |
|
9 |
I2 / 0.25 |
1.5 |
1,5 |
90 |
67.5 |
23.7 |
8.8 |
– |
|
10 |
I2 / 0.2 |
1.2 |
1,5 |
70 |
43.8 |
10.3 |
13.4 |
32.5 |
|
11 |
I2 / 0.2 |
1.2 |
1,5 |
90 |
64.8 |
14.3 |
14.3 |
6.6 |
|
12 |
I2 /0.6 |
0 |
3 |
90 |
– |
– |
– |
100 |
The maximum content of iodopentafluorobenzene in the reaction products, which we could reach by changing of reagents and temperature, is 25 % (experiment № 5). The main product of the reaction in all variants was chloropentafluorobenzene. It was also shown (table 1), that for complete conversion of starting pentafluorobenzene for 2 hours, 3 equivalents of chlorosulfuric acid are needed by the reaction within 60 0 – 70 0C. The content of chlorosulfuric acid can be reduced when performing the process by higher temperature (900C). It was also found out, that by using thionyl chloride as dehydrating agent, it was also possible to reduce the quantity of chlorosulfuric acid to 1.5 moles, and moreover the higher temperature is also needed (900C). Taking into account the obtained data, minimum 0.15 moles of iodine are needed to reach the complete conversion of pentafluorobenzene. The application of iodine chloride instead of elemental iodine results in the same products.
When using iodine bromide, bromopentafluorobenzene with yield 97 % is produced. Chloropentafluobenzene is produced as an impurity (2.8 %) (Scheme 2).
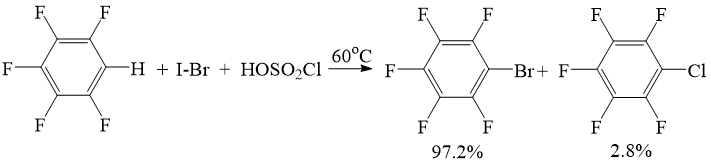
Scheme 2
It was assumed, that the reaction goes through intermediate formation of pentafluorobenzene sulfochloride (according to scheme 3), because the method of preparation of sulfochlorides through direct interaction between chlorosulfuric acid and pentafluorobenzene by the heating [8]. Perfluoroalkyl halides preparation from perfluoroalkyl sulfochlorides is also known [9].
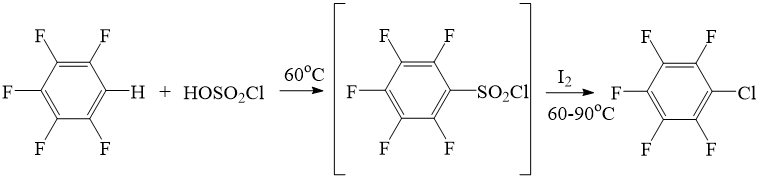
Scheme 3
In fact it appeared that, sulfochloride didn’t react with iodine neither in the absence, nor in the presence of chlorosulfuric acid both by temperature 60 0C, and by higher temperature (till 900C) , and it was completely returned from reaction unchanged.
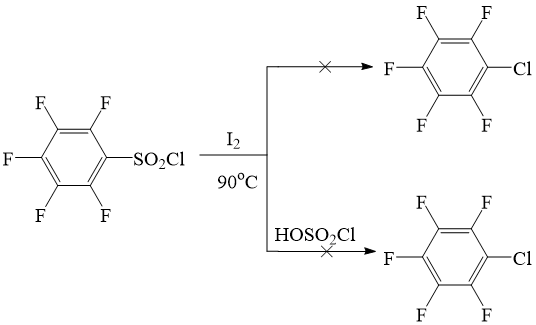
Scheme 4
Based on the above, the mechanism of this reaction and how the substitution of fluorine atom in the ortho-position with formation of 1,2-dichlorotetrafluorobenzene takes place, still remain a riddle for us.
Experimental
Typical method for obtaining of chloropentafluorobenzene:
In a 250 ml two-neck flask, provided with thermometer, dropping funnel and reflux connected on exit to a trap, which is cooled with the mixture of dry ice and acetone, 34.8 g (0.214 mol) of iodine chloride and 30.0 g (0,178 mol) of pentafluorobenzene are added to 62.4 g (0.535 mol) of chlorosulfuric acid, while stirring. The reaction mass is kept during 2 hours by 70oC. The reaction is considered to be complete, based on growth of volume of sulfur (IV) oxide in the trap. The reaction mass is cooled by room temperature and poured into 300 g of crushed ice, while stirring 15 % solution of NaHSO3 is dropped till decolouring, lower layer is washed with 2 х 30 ml water, dried under CaCl2, then it is filtered and distilled. It is obtained 39 g of the mixture, which according to GC contains 73% of chloropentafluorobenzene, 6.8% of 1,2-dichloropentafluorobenzene and 10.2% of iodopentafluorobenzene (ref. table № 1, experiment № 1).
Method for obtaining of Bromopentafluorobenzene
In a 250 ml two-neck flask, provided with thermometer, dropping funnel and reflux, connected on exit to a trap, which is cooled with the mixture of dry ice and acetone, 44,3 g (0,214 mol) of iodine bromide and 30,0 g (0,178 mol) of pentafluorobenzene are added to 62,4 g (0,535 mol) of chlorosulfonic acid by 20 0C, while stirring. The reaction mass is kept during 2 hours by 600C. The reaction is considered to be complete, based on growth of volume of sulfur (IV) oxide in the trap. The reaction mass is cooled by room temperature and poured into 300 g of crushed ice, while stirring 15 % solution of NaHSO3 is dropped till decolouring, lower layer is washed with 2 х 30 ml water, dried under CaCl2, then it is filtered and distilled. It is obtained 38 g of mixture, which according to GC contains 97,2 % bromopentafluorobenzene and 2,8 % of chloropentafluorobenzene.
References
- V.A.Montes, G.Li, R.Pohl, Adv.Mater., 2004, v.16, 2001-2003.
- M. Matsui, K. Shirai, N. Tanaka, K. Funabiki, H. Muramatsu, K. Shibata, Y. Abe and Y. Ohgomori, J.Mater.Chem., 1999, v.9, p.2755-2763.
- W. Pummer, L. Wall, Res. NBS, 63A,167 ,1959.
- E. Nield, R. Stephens, J. Tatlow, J. Chem. Soc.,1959, p.166.
- N.N. Vorozhcov ml., V.A. Barhash, N.G. Ivanova, S.A.Anichkina, O.I. Andreevskaya, DAN SSSR, 159,125, 1964.
- I. Popov, H.-Q. Do, and O.Daugulis, Journal of Organic Chemistry, 74(21), p.8309-8313; 2009.
- Rodrigo S. Da Ribeiro, Pierre M. Esteves, Marcio C.S. de Mattos; Synthesis; 2011, №5, p. 0739-0744.
- Sintezy ftororganicheskih soedinenij (Pod red. I.L. Knunyantsa i G.G.Yakobsona), Moskva, izd-vo "Khimiya", 1973, 312 s., s.189.
- US Patent 7692048 (2010)
Recommended for publication by Prof. S. M. Igumnov
Fluorine Notes, 2017, 112, 5-6
