Received: December, 2016
DOI 10.17677/fn20714807.2016.06.03
Fluorine Notes, 2016, 109, 5-6
Original Method of Synthesis of Pentafluoroethyl Iodide
V.E. Boikoab, A.V. Edunovb, S.М. Igumnovab
aA.N. Nesmeyanov Institute of Organoelement Compounds, Russian Academy of Sciences, Russia, 119991, GSP-1, Moscow, V-334, Vavilova St, 28
bZAO NPO PiM-INVEST, Russia, 119334 Moscow, Leninsky prospect 4
e-mail: boykii@mail.ru
Abstract: Perfluoroethyl iodide is a much required compound for obtaining of different classes of organic compounds. We developed a new, original method of synthesis of pentafluoroethyl iodide from perfluoropropylene.
Keywords:perfluoroethyl iodide, perfluoropropylene, 1,1,1,2,3,3-hexafluoro-2-iodo-3-methoxypropane.
Perfluoroethyl iodide is widely applied for the introduction of perfluoroethyl group into compounds of different classes, through its addition to the double bond of unsaturated compounds [1], so that it is used for obtaining of partially fluorinated alcohols, esters, acids, silanes, and other functional derivatives, which are applied for the synthesis of medical substances, surface-active materials etc.
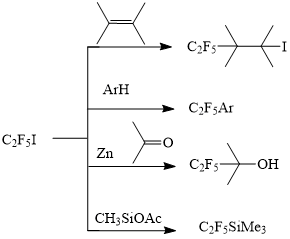
For example Pentafluoropentanol, which is produced in commercial quantities through addition of perfluoroethyl iodide to allyl alcohol, with the following reduction of iodine by hydrogen on catalyst [2] , is an intermediate for the synthesis of complicated biologically active compounds, mainly steroid drugs for the treatment of cancer diseases [3.4]
Ketogroup addition of perfluoroethyl iodide is another method of introduction of pentafluoroethyl fragment into different complicated molecules [5]. There are a lot of examples of obtaining of different pentafluoroethylaryl derivatives from perfluoroethyl iodide [6.7].
Perfluoroethyl iodide is also used as telogen in the reaction with tetrafluoroethylene for the obtaining of higher iodides [8].
Perfluoroethyl iodide also reacts by radical mechanism with aromatic compounds, giving perfluoroethyl derivatives of aromatic compounds [9,10]. Besides, perfluoroethyl aromatic compounds can be obtained not only from aryl iodides, but from aryl compounds, without iodine.
There is also a known method of obtainment of perfluoroethyl iodide by reaction of tetrafluoroethylene with iodine pentafluoride (IF5), [11]. The disadvantage of this method is difficulty of transportation of perfluoroethylene, which makes this method available only for manufacturers of perfluoroethylene.
Another known method of obtainment of perfluoroethyl iodide is decomposition of silver salt of heptafluoropropionic acid in the presence of iodine [12]. There is a known method of obtainment of perfluoroethyl iodide by decomposition of potassium salt of perfluoropropionic acid in dimethylformamide. [13] The disadvantage of this method is expensiveness of starting materials.
We developed a three-stage method of synthesis of pentafluoroethyl iodide from perfluoropropylene. On the first stage, as the result of reaction of conjugate addition, the product 2 was obtained, which then through defluoromethylation with catalytic amount of antimony pentachloride resulted in the formation of acid fluoride 3. The following decarbonylation in the presence of catalytic amount of anymony pentafluoride resulted in formation of target product 4 (Scheme 1).
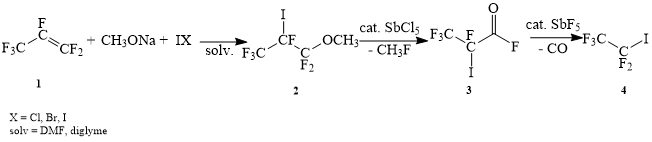
Scheme 1
Since the stage of obtainment of acid fluoride 3 and its following conversion into pentafluoroethyl iodide didn’t cause any special difficulties and the yield on every stage was more than 90 %, our main efforts were concentrated on the first stage of obtainment of 1,1,1,2,3,3-hexafluoro-2-iodo-3-methoxypropane.
Initially, we tried to obtain 1,1,1,2,3,3-hexafluoro-2-iodo-3-methoxypropane according to the sheme 2. But the main product of this reaction was the product of addition of methanol to the double bond of propylene 5.
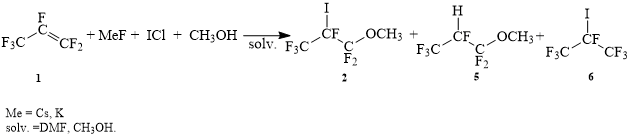
Sheme 2.
Taking into account the obtained results, it was decided to use sodium methylate as the source of alkoxide-anion.
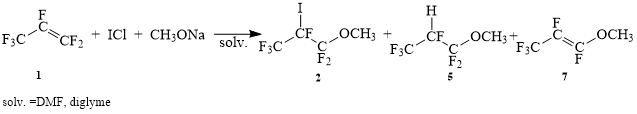
Sheme 3.
It was indicated in the table1, that the quality of used sodium methylate was very important. Experiments 1,2 was performed with purchased methylate, as a result the main product was product 5. In the reactions 3-7, fresh sodium methylate was used. It was also shown, that the nature of counterion in the source of I+ didn’t much influence the percentage of reaction products. It was not found out the influence of solvent and sequence of addition of reagents on content of reaction products. It was also shown, that the quantity of used in the reaction sodium methylate resulted only in the increase of reacting perfluoropropylene, but it didn’t influence the percentage of reaction products.
Table 1.
# |
IX(eq.) |
CH3ONa(eq.) |
Solv. |
CF3CFICF2OCH3 |
CF3CFHCF2OCH3 |
CF3CF=CFOCH3 |
1 |
ICl |
2 |
DMF |
22.5% |
66.1% |
1.5% |
2 |
ICl |
1.2 |
DMF |
30.8% |
54.6% |
1.4% |
3 |
ICl |
1.2 |
DMF |
51.2% |
26.8% |
3.9% |
4 |
ICl |
1.2 |
DMF |
41.4% |
32.3% |
6.3% |
5 |
ICl |
3 |
DMF |
27.6% |
30.6% |
15.5% |
6 |
IBr |
1.5 |
Diglyme |
42.5% |
14% |
8.3% |
7 |
IBr |
3 |
DMF |
42% |
21% |
9.5% |
Experimental
Obtaining of 1,1,1,2,3,3-hexafluoro -2-iodo-3-methoxypropane (2).
Method #1
In a four-neck flask 200 g of methanol are added. 64.9 g (0.399 mol) of iodine chloride are dropped at a temperature 0°С though dropping funnel. Meanwhile the reaction mass is warmed to 5 – 10 °С. Then portionwise at temperature 0°С fresh annealed caesium fluoride (111.46 g 0.733 mol) or fresh annealed potassium fluoride (42.53 g 0.733 mol) are dropped, meanwhile the reaction mass is warmed to 5°С. After adding the whole quantity of CsF or KF, the reaction mass is stirred at temperature 10-15 °С during 0,5 hours, then during 2 hours 50 g (0.333 mol) of hexafluoropropene are bubbled at temperature 5- 10°С. Upon completion of feeding, the reaction mass is stirred during 1 hour. Then it is poured into equal volume of 15 % hydrochloric acid, the lower layer is separated, washed with 5 % aqueous solution of NaHSO2 and Na2S2O3 till decolouring. It is obtained 30 g of crude, containing: 10 % of heptafluoroisopropyl iodide 6; 10 % of methoxy iodide 2 ; 50 % of methoxy ester 5.
Method # 2
Method # 2 is analog to Method # 1
m (perfluoropropylene)=50 g (0.333 mol); m (ICl)= 64.9 g (0.339 mol); m (CH3ONa)= 26.9 g (0.4999 mol), V (DMF or diglime)= 150 ml
The replacement of sequence of addition of reagents didn’t influence the content of final substances. It is obtained: 64 g of crude, containing: 41.4% 2, 32.3% 5, 6.3% 7.
Method # 3
Method # 3 is analog to Method #1. Iodine bromide is prepared directly in the still, through solution of calculated quantity of iodine in DMF and dropping of bromine. m (M-6)= 50 g (0.333 mol); m (IBr)= 82.7 g. (0.339 mol); m (CH3ONa)= 26.9 g. (0.4999 mol); V (DMF or diglime) = 150 ml. It is obtained 44 g of crude, containing: 52% 2, 20% 5, 10% 7.
Obtainment of 2,3,3,3-tetrafluoro-2-iodopropionyl fluoride (3).
In a 3-neck flask, to 50 g (0.162 mol) of methoxy iodide 2 , 4.4 g (0.0162 mol) of antimony pentachloride are dropped with such speed, so that moderate gassing is observed. After dropping of the whole quantity of SbCl5 , the reaction mass is boiled during 0.5 hours, till gassing is stopped. Meanwhile the temperature in still is 63 °С. Then reflux is replaced with fractional column with Liebig condenser, alonge and receiving flask, which is connected on exit with Tischenko flask with H2SO4. Then it is distilled. It is obtained 40 g of acid fluoride 2. Boiling point = 58-59 °С. Yeild: 90 %
Obtainment of 1,1,1,2,2-pentafluoro-2-iodoethane (4).
In a 3-neck flask, to 40 g (0.1459 mol) of acid fluoride 3 , while stirring at temperature 40 °С 34.8 g (0.1606 mol) of antimony pentafluoride are added dropwise with such speed, so that moderate gassing is observed, with simultaneous distillation of ethyl iodide, which is bubbled in DMF. The solution of ethyl iodide with DMF is dropped in 30 % hydrochloric acid, with simultaneous distillation of ethyl iodide into receiving flask, which is cooled by mixture of dry ice and acetone. It is obtained 32.3 g of pentafluoroethyl iodide 4. Boiling point = 12- 13 °С. The yield : 90 %
References
- N.O.Brace, J Org.Chem., 1962, 27, 4491, 3033
- US6002053, 1999.
- US5502046, 1996.
- WO2005077968.
- US2012190660.
- US2014221435.
- EP2319830, 2011.
- Sosnovsky, Macmillan, Free radical reactions in Preparative Organic Chemistry, New York.
- US2014221435, 2014.
- EP2319830, 2011.
- US2614131, 1949.
- J Chem. Soc., 1953, 3761.
- Paskovich D., Gaspar P., Hammond G.S., J Org.Chem., 1967, 833.
Recommended for publication by Prof. S. Igoumnov
Fluorine Notes, 2016, 109, 5-6
