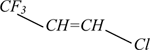Received: December, 2014
DOI 10.17677/fn20714807.2015.02.02
Fluorine Notes, 2015, 99, 3-4
PROCESSES FOR THE MANUFACTURE OF 1-CHLORO-3,3,3-TRIFLUOROPROPENE
N.N. Zatsepina, N. V. Peganova, V.G. Barabanov, I.G. Trukshin, A.A. Zhukov
Federal State-owned Unitary Enterprise "Russian Scientific Center "Applied Chemistry", 193232, St.Petersburg, ul. Krylenko, 26A, e-mail: vbarabanov@mail.ru
Abstract: Available methods for the manufacture of trans–1-chloro-3,3,3-trifluoropropen (1233zd (Е)) are analyzed, among them the formation of intermediate (1233zd (Е+Z)) isomeric blend, and the methods for cis→ trans isomerization are considered as well.
Keywords: 1-Chloro-3,3,3-trifluoropropene, 1,1,1,3,3-pentafluoropropane, liquid-phase hydrofluorination, gas-phase hydrofluorination.
Introduction
1-Chloro-3,3,3-trifluoropropene is classified with hydrochlorofluorolefins. Currently in industry it is applied for solvent, foaming agent, refrigerant, or component of refrigerants or solvent compositions. It is also used for a monomer in the synthesis of polymers or other fluorinated materials.
The substance is a promising candidate for the substitution of 1,1,1,3,3-pentafluoropropane (HFС-245fa) that in spite of its favorable properties is not recommended for numerous applications because of considerably high global warming potential (GWР).
1-Chloro-3,3,3-trifluoropropene has similar technical properties, but very low ozone-depleting potential (ODР ~0) and low GWР (<5).
Product properties:
|
Chemical name |
1-chloro-3,3,3-trifluoropropene (HCFO–1233zd or 1233zd); |
|
|
Molecular formula |
C3H2ClF3 |
|
|
Structural formula |
CF3CH=CHCl |
|
|
Molecular mass |
130,5 |
HCOF–1233zd has 2 stereoisomers:
|
– |
trans–isomer (main isomer): |
||
|
Chemical name |
trans–1-chloro-3,3,3-trifluoropropene (1233zd (Е)); |
||
|
CAS |
2730–43–0 |
||
|
Molecular formula |
C3H2ClF3 |
||
|
Structural formula |
|
||
|
Boiling point |
21°С at 760 mm Hg |
||
|
Melting temperature |
|
minus 10°С |
|
|
Density |
|
1,312g/cm3 |
|
|
Refraction index |
|
1,348 |
|
| |
|||
|
– |
cis–isomer: |
||
|
Chemical name |
|
cis–1-chloro-3,3,3-trifluoropropene (1233zd (Z)); |
|
|
CAS |
|
99728–16–2 |
|
|
Molecular formula |
|
C3H2ClF3 |
|
|
Structural formula |
|
|
|
|
Boiling point |
38°С at 760 mm Hg |
||
In most applications they use trans–isomer (1233zd (Е)) or blend of both isomers (1233zd (Е+Z)). The methods for the manufacture of individual isomers were developed for the applications where physical difference between those two isomers proved to be essential.
Below reviewed are the chief processes for the manufacture of trans–1-chloro-3,3,3-trifluoropropene (1233zd (Е)) including those with intermediate formation of (1233zd (Е+Z)) mixture, and the methods for cis-trans isomerization.
Methods for the manufacture of 1-chloro-3,3,3-trifluoropropene
For the production of trans–1-chloro-3,3,3-trifluoropropene (1233zd (Е)) they usually make use of liquid-phase or gas-phase reactions between anhydrous hydrogen fluoride and 1,1,1,3,3-pentachloropropane (CCl3CH2CHCl2; HCС-240fa), 1,3,3,3-tetrachloropropene (CCl3CH=CHCl), 1,1,3,3-tetrachloropropene (CНCl2CH=CCl2), or mixture thereof in the presence of a corresponding catalyst (see, e.g.[1-8]; ibid. the earlier references). In recent years non-catalytic liquid-phase processes for the manufacture of isomer 1233zd (Е) were being developed on the basis of HCС-240fa (see, e.g. [9-12]).
The mechanism of all above processes involves one-step reactions, e.g., that of 1,1,1,3,3-pentachloropropane (HCС-240fa) with anhydrous hydrogen fluoride in gas or in liquid, resulting mostly in the formation of trans–1-chloro-3,3,3-trifluoropropene (1233zd (Е)) and by-produced HCl. Preferably, the process conditions (temperature, pressure, contact time) are maintained so that to increase the (Е)/(Z)–isomer ratio, and to minimize interacting of anhydrous hydrogen fluoride with thus formed 1233zd (Е) that would result in HСFС-244fa. The latter may further react resulting in HFO–1234zе.
Following the above the «useful» reactions involve:
|
CCl3CН2CHCl2 + 3HF |
→ |
CF3CH=CHCl + 4HCl |
|
1,1,1,3,3-pentachloropropane |
1233zd (Е) |
|
|
CCI3CH=CHCl +3HF |
→ |
CF3CH=CHCl + 3HCl |
|
1,3,3,3-tetrachloropropene |
1233zd (Е) |
|
|
CНCl2CH=CCl2 +3HF |
→ |
CF3CH=CHCl + 3HCl |
|
1,1,3,3-tetrachloropropene |
1233zd (Е) |
|
CCl3CН2CHCl2 + 3HF |
→ |
CF3CH=CHCl + 4HCl |
||
|
1,1,1,3,3-pentachloropropane |
1233zd (Z) |
|||
|
CF3CH=CHCl + HF |
→ |
CF3CH2СHFCl |
||
|
(Е) or (Z) 1233zd |
244fa |
|||
|
CF3CH2СHFCl + HF |
→ |
CF3CH=CHF + HCl |
||
|
244fa |
1234zе |
|||
A number of methods for the manufacture of 1233zd (Е) were based on catalytic dehalogenation of 1,1,1-trifluoro-3,3-dichloropropane (НСFС–243fa) [13] or 1,1,1,3-tetrafluoro-3chloropropane (НСFС–244fa) [14]:
![]()
![]()
1. Catalytic liquid-phase process for the manufacture of 1-chloro-3,3,3-trifluoropropene [1, 3-8]
The process is based on a catalyzed liquid-phase reaction of hydrogen fluoride with HCС-240fa or HCС-240fa plus 1,1,3,3-tetrachloropropene and/or 1,3,3,3-tetrachloropropene, and results in the production of target 1233zd (Е) and some by-products (HCl, 1233zd (Z), etc.) further separated with the help of corresponding scrubbing systems.
The liquid-phase process for 1233zd (Е) manufacture involves 6 main steps:
- Catalyst preparation (TiCl4–based catalyst is preferable). Fluorination catalysts other than TiCl4 are also applicable, those being SnCl4, TaCl5, SbCl3, FeCl3 or AlCl3, that partially or totally fluorinated by hydrogen fluoride.
- Fluorination (periodical, semicontinuous, continuous) by anhydrous hydrogen fluoride combined with withdrawal of mixture containing chiefly 1233zd (Е+Z), by-produced HCl, and НF (in azeotropes) from the reactor.
- Isolation and purification of by-produced HCl.
- Isolation and recycling into the reactor of surplus НF.
- Purification of the target product, 1233zd (Е).
- Isomerization of by-produced 1233zd (Z) to result in 1233zd (Е).
Those procedures are illustrated in Fig.1–4 with a number of layouts for continuous liquid-phase manufacture of 1233zd (Е) shown.
Procedure 1:
Catalyst is fluorinated in a stirred reactor at 0–120°С. The degree of catalyst fluorination, partial or exhaustive, is estimated by the liberation of by-produced HCl that increases the reactor pressure.
Procedure 2:
Fluorination is conducted in a liquid-phase reactor equipped with a separation column for light fractions topping. It is preferable to use reactors made of materials resistant to corrosion action of hydrogen fluoride such as Hastelloy, Inconel, Monel, or fluoropolymer-coated vessels. The reactor must be provided with a stirrer, heat-exchange jacket and temperature- and pressure- controls.
With the help of an external heat-exchanger the separation column temperature is maintained 10–40°С below the average reactor temperature. Gaseous fluorination products passing the separation column contain the target 1233zd (Е+Z), by-produced HCl, low-boiling organic by-products, e.g., 1234zе (Е+Z), and considerable quantity of anhydrous hydrogen fluoride (in azeotropes). Thanks to the separation column most of hydrogen fluoride, original reagents, partially fluorinated products, dimeric products and fluorinated catalyst remain in the reactor. Usually the liquid-phase fluorination process is conducted under pressure 80–140psig (~5.5–10atm) at 85–120°С (preferably at 95–100°С, as this regime favors the maximal formation of 1233zd (Е) as compared to 1233zd (Z)).
Procedure 3:
Isolation of HCl is conducted in a low-temperature distillation column at temperature below minus 40°С. HCl passing the column top may be absorbed by deionised water resulting in concentrated HCl or used in some other chlorinated products manufacture.
Procedure 4:
After HCl separation crude product in the still of low-temperature separation column consists mainly of 1233zd (Е+Z) and НF (sometimes up to 30 mass.%). Separation of hydrogen fluoride from is usually either extracted from organic phase with sulfuric acid in a sulfuric-acid extractor or separated at a low-temperature separator. After additional rectification НF is recycled into the reactor.
Procedure 5:
To remove hydrogen fluoride traces organic phase is then passed through an absorber filled with water or alkaline solution, and then through an adsorber charged with a drier (zeolite 3А, silicagel, aluminium oxide, etc.). The product finish purification is conducted in two (or more) distillation columns. This allows isolation of both 1233zd stereoisomers (1233zd (Е) Тboiling=21°С и 1233zd (Z) Тboiling~38°С).
Procedure 6:
When trans–isomer is the target product its yield may be increased through effective cis→trans isomerization of cis–isomer in a gas-phase reactor in the presence of a catalyst (preferably, fluorinated chromium oxide) at 50–350°С.
Figure 1 shows the layout of continuous liquid-phase process for the manufacture of 1233zd (Е) involving sulfuric-acid separation of hydrogen fluoride from the system. Here, at first, fluorinated catalyst is prepared at 0–120°С in a reactor (pos.R–1) by treatment of some metal chloride catalyst with excessive amount of anhydrous hydrogen fluoride (e.g., with TiCl4, the corresponding mole ratio НF:TiCl4 > 4:1). НCl released during the catalyst preparation is removed from the top of the separation column (pos.СS–1), while the excessive reactor pressure is under permanent control. When the catalyst preparation is over additional amount of hydrogen fluoride and 1,1,1,3,3-pentachloropropane and/or 1,1,3,3-tetrachloropropene, and/or 1,3,3,3-tetrachloropropene is introduced into the reactor under thorough stirring.
Fluorination starts when the mixture is heated to 85°С. Then blend of chlorinated products with HF (at stoichiometric ratio) is being continuously fed through a heat-exchanger (pos.НХ–1) into the reactor (pos.R–1). The chlorinated product blend may also be fed into the reactor without heat-exchanger (pos.НХ–1). Work pressure 60–160psig (~4–11atm) or, preferably, 80–140psig (~5,5–10atm) is maintained with the help of a valve at the column top (pos.СS–1). Work temperature 85–120°С is controlled with the help of the heat-exchanger jacket. The separation column (pos.СS–1) provides recycling of catalyst particles, НF, partly fluorinated substances and non-reacted original reagents into the reactor for further processing.
Gas flow passing the separation column top (pos.СS–1) consists mainly of 1233zd (Е+Z), НCl, and НF (and small amount of partly fluorinated intermediates and by-products) is directed into a low-temperature separation column (pos.D–1), where gaseous НCl is separated. Thus separated НCl may be utilized.
The column (pos.D–1) stills, containing mainly 1233zd (Е+Z) and НF is directed into a system intended for hydrogen fluoride separation. At first after evaporation in the heat-exchanger (pos.НХ–2) 1233zd/HF flow enters НF–absorption column (pos.А–1). Here 1233zd/НF gas flow in counterflow contacts liquid flow of 50–80% sulfuric acid that absorbs most of НF. Then НF/H2SO4/H2O still flow from column (pos.А–1) is directed into a heat-exchanger (pos.НХ–3) to be heated to temperature sufficient for separation of most of НF from H2SO4/H2O mixture. The flow then enters НF–regeneration column (pos.D–3). After НF separation in the heat-exchanger (pos.НХ–3) the flow consists mainly of H2SO4 and H2O (with 0–2% НF), and after cooling in a heat-exchanger (pos.НХ–4) it is recycled into the absorption column (pos.А–1). Still residue of H2SO4/H2O after НF regeneration in column (pos.D–3) pours into the heat-exchanger (pos.НХ–3). Regenerated НF is returned into the reactor (pos.R–1) through the evaporator (pos.Н–1).
Leaving the top of the absorption column (pos.А–1) gas flow consists mainly of 1233zd (Е+Z) with HF traces. To remove HF the flow firstly directed into a scrubbing system (pos.А–2) where it contacts with aqueous or alkaline solution, and then into adsorber charged with drier. Acid-free product enters the first of two rectification columns (pos.D–4) to separate «light» by-products with boiling points below that of 1233zd (Е). The flow from the column still (pos.D–4) containing mainly 1233zd (Е), 1233zd (Z), and «heavy» fluorinated by-products is directed into the second distillation column (pos.D–5) where pure marketable 1233zd (Е) is distilled off to be directed then to storage, then 1233zd (Z) is distilled off from the residuum, and via evaporator (pos.НХ–5) directed into isomerization reactor (pos.R–2), where 1233zd (Z) is converted mostly to 1233zd (Е). The flow leaving reactor (pos.R–2) is recycled for purification into column (pos.D–4). In general, any by-products formed in reactor (pos.R–2) may be unstable and decompose to give НF and НCI. In this case the flow from reactor (pos.R–2) is directed into scrubbing system (pos.А–2) to remove acidic impurities. Stillage from column (pos.D–5) may be recycled into reactor (pos.R–1). Gummy stillage is directed into scrubbing systems.
The layout for 1233zd (Е) manufacture in Fig.2 differs from that of Fig.1 by an additional recycling column (pos.D–2) installed between separation column (pos.СS–1) and НCl-distilling column (pos.D–1) in order to increase the purification process efficiency. The temperature regime of column (pos.D–1) allows to clean gas flow from separation column (pos.СS–1) of most of fluorinated impurities while keeping blend of 1233zd (Е+Z), HCl and и НF. Stillage residue from column (pos.D–1) is recycled into reactor (pos.R–1).
The layouts for 1233zd (Е) manufacture in Fig.3, 4 differ from those in Fig.1, 2, correspondingly only by a phase separator provided for recycling hydrogen fluoride from 1233zd/НF blend.
Having this in mind the still flow consisting mainly of 1233zd (Е+Z)/НF blend is firstly directed from column (pos.D–1) (Fig.3) into heat-exchanger (pos.НХ–2) to be cooled to temperature below 0°С, and then into phase separator (pos.RS–1) where temperature ranges between minus 40°С and minus 5°С. НF-enriched upper layer (10% 1233zd) is recycled into liquid-phase reactor (pos.R–1). Organic-enriched lower layer, containing mainly 1233zd (4% НF) is directed into scrubbing system (pos.А–1) via evaporator (pos.НХ–3).
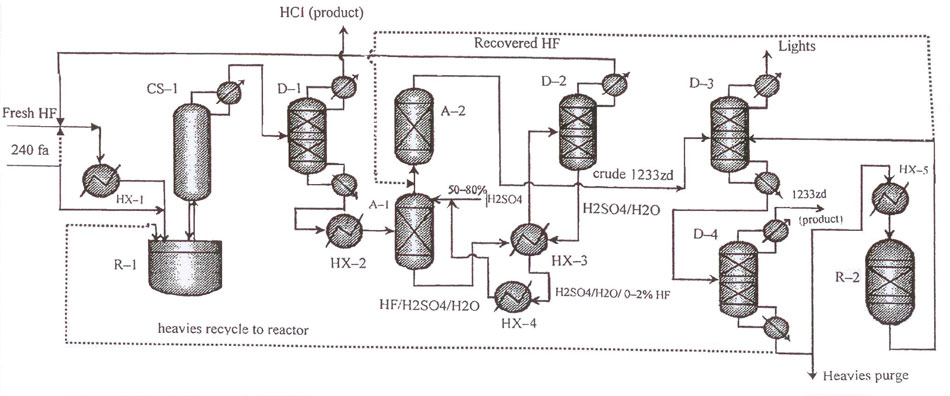
Fig.1. Layout for continuous catalytic liquid-phase process for 1233zd (E) manufacture with sulfuric acid-based
variant of hydrogen fluoride isolation from HF/HCFO-1233zd mixture [3].
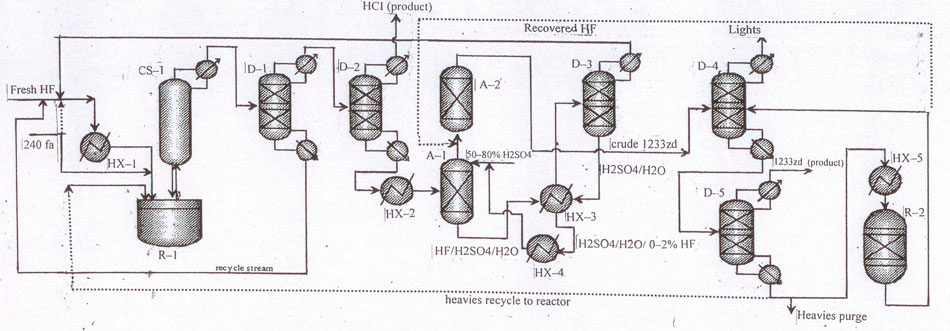
Fig.2 Layout for continuous catalytic liquid-phase process for 1233zd (E) manufacture with sulfuric acid-based variant of hydrogen fluoride isolation from HF/HCFO-1233zd mixture. For more efficient purification of the target product a column for return products separation (pos.D-1) is added to the layout.
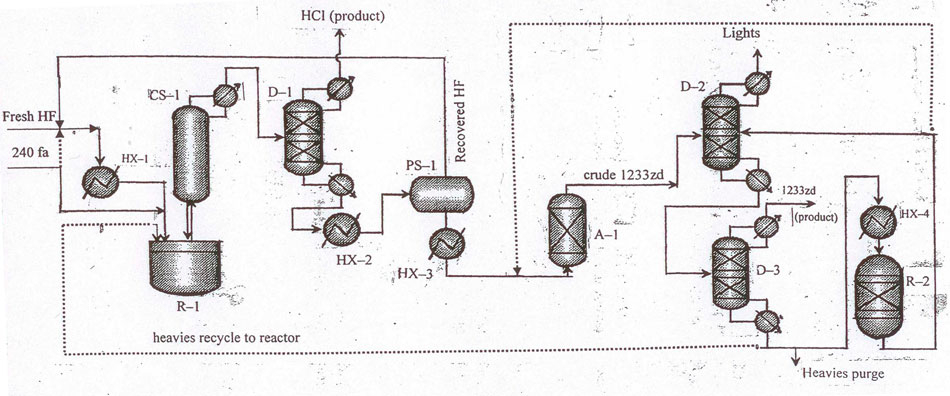
Fig.3 Layout for continuous catalytic liquid-phase process for 1233zd (E) manufacture with hydrogen fluoride isolation from HF/HCFO-1233zd mixture in a phase separator.
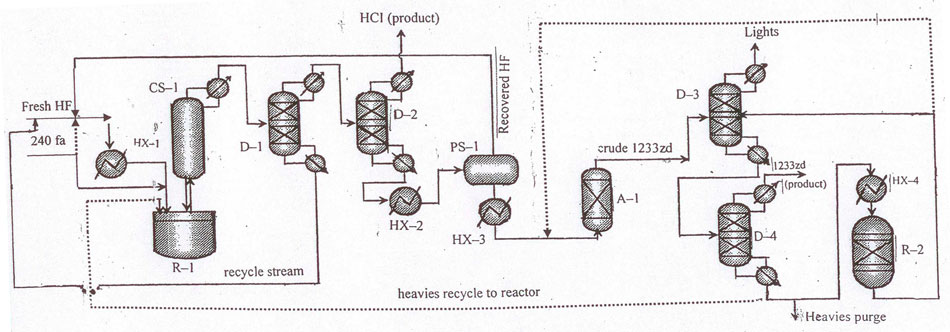
Fig.4 Layout for continuous catalytic liquid-phase process for 1233zd (E) manufacture with hydrogen fluoride isolation from HF/HCFO-1233zd mixture in a phase separator. To increase the efficiency of the target product purification a column for return products (pos.D-1) is added to the layout.
2. Catalytic gas-phase process for the manufacture of trans-1-chloro-3,3,3-trifluoropropene [2-5]
The process uses a catalytic gas-phase reaction between НF and НСС–240fa resulting in the formation of target 1233zd (Е) and by-produced (HCl, 1233zd (Z), etc.), which are separated with the help of scrubbing systems similar to those described above for 1233zd (Е) liquid-phase manufacture process.
The process for 1233zd (Е) gas-phase manufacture involves 5 main steps:
- НСС–240fa continuous fluorination with anhydrous hydrogen fluoride, and simultaneous withdrawal from the reactor of mixture containing thus formed 1233zd (Е+Z), by-produced НCl, impurities, and, partially, hydrogen fluoride.
- Isolation and purification of by-produced НCl.
- Surplus hydrogen fluoride separation and recycling into the reactor.
- Purification of target 1233zd (Е) and recycling of high-boiling fluorinated products into the reactor.
- Isomerization of by-produced 1233zd (Z) to 1233zd (Е) in order to increase its yield.
Procedure 1:
Any gas-phase fluorination catalysts are generally applicable in the fluorination process. The available catalysts are based on chromium, aluminium, cobalt, manganese, nickel, or iron compounds, including oxides, hydroxides, halogenides, oxyhalides, inorganic salts, or blends thereof, supported or not. Gas-phase catalysts may also include some combinations of well-known catalysts, e.g., Cr2O3/Al2O3, Cr2O3/AlF3, Cr2O3/C, CoCl2/Cr2O3/Al2O3, NiCl2/Cr2O3/Al2O3, CoCl2/AlF3, NiCl2/AlF3, etc. The catalysts based on FeCl3/C, SnCl4/C, TaCl5/C, SbCl5/C, AlCl3/C and AlF3/C are also applicable. Aluminium oxide or aluminium oxyfluoride are applicable for support of metal halide catalysts.
Fluorinated chromium oxide is the most widely applied catalyst.
For Cr2O3–based catalysts both crystal and amorphous chromium oxide is applicable. Amorphous form is easier available commercially, and hence, its usage is more preferable. The catalyst purity must be at least 98%. The catalyst amount must be sufficient for the realization of fluorination, but excessive quantity is also allowed. The catalyst performance depends also on its grain size.
Any type of gas-phase reactors is applicable for fluorination process. Preferable are the reactors made of materials resistant to corrosion effect of hydrogen fluoride and catalysts, among those are Hastelloy–С, Inconel, Monel, Incalloy or steel vessels coated with fluoropolymers.
In figures 5–8 various layouts for continuous gas-phase process of 1233zd (Е) manufacture are shown that differ from liquid-phase processes shown in figs 1–4 only by the fact that fluorination is conducted in a gas-phase reactor.
A gas-phase reactor (pos.10 Figure 5) is charged with catalyst (preferably, fluorinated chromium oxide). At 200-450°С (preferably 275–375°С) under pressure 0.15-7atm НF/НСС–240fa blend is continuously fed into the reactor via an evaporator (pos.1). Molar ratio НF: НСС–240fa is at least 3:1, but, preferably, between 5:1 and 11:1.
The flow leaving the reactor involves 1233zd (Е+Z), partly fluorinated by-products, НCl, and НF. Further procedures for 1233zd (Е) isolation and purification are similar to those described above for liquid-phase process.
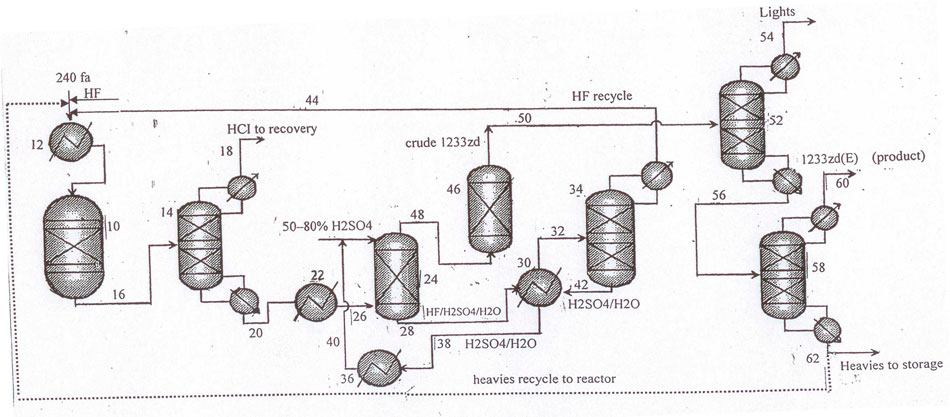
Fig. 5 Layout for continuous catalytic gas-phase process for the manufacture of 1233zd (E) with sulfuric acid-based variant of hydrogen fluoride isolation from HF/HCFO-1233zd mixture.
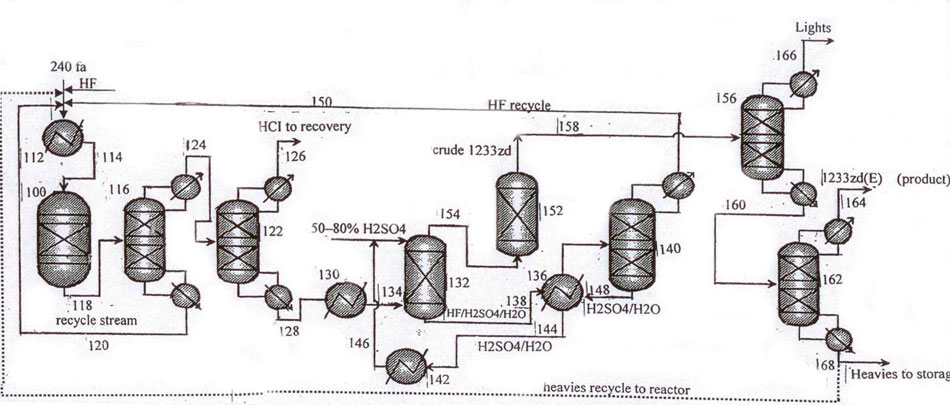
Fig.6 Layout for continuous catalytic gas-phase process for the manufacture of 1233zd (E) with sulfuric acid-based variant of hydrogen fluoride isolation from HF/HCFO-1233zd mixture. Recycling column (pos.116) is introduced additionally for more efficient purification of the target product.
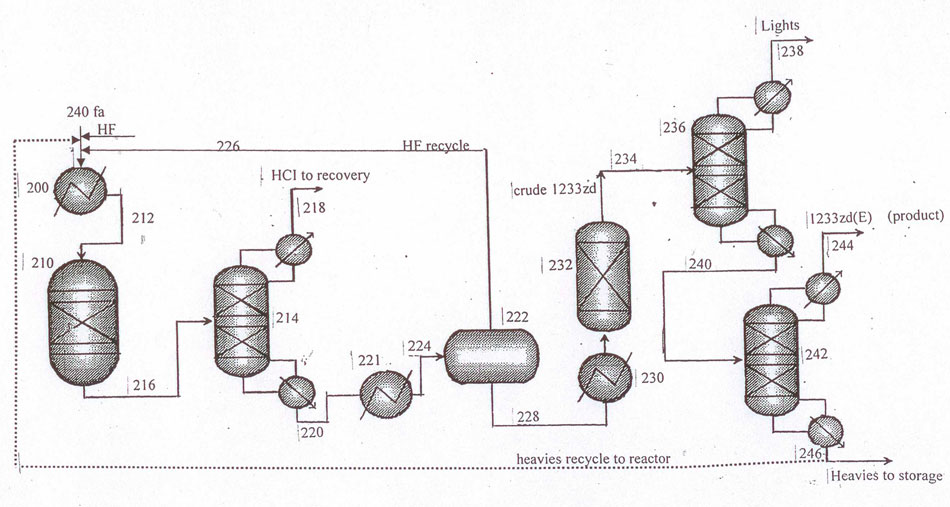
Fig.7 Layout for continuous catalytic gas-phase process for the manufacture of 1233zd (E) with sulfuric acid-based variant of hydrogen fluoride isolation from HF/HCFO-1233zd mixture in a phase separator.
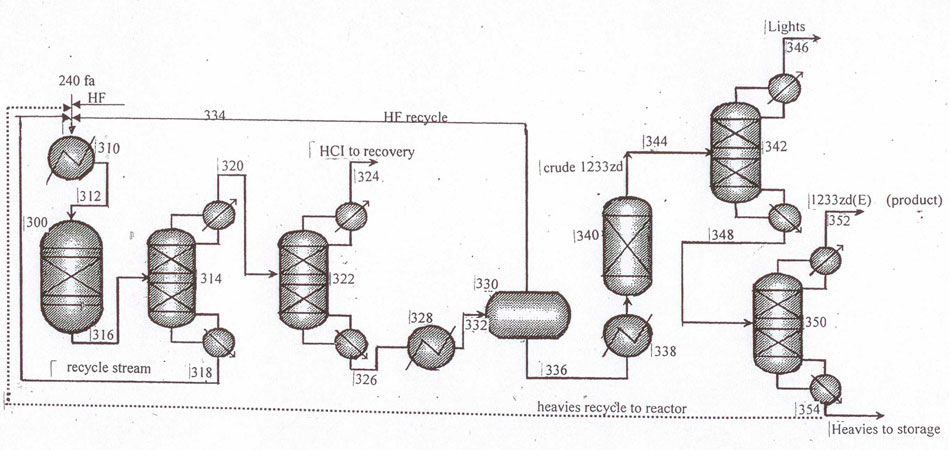 Fig.8 Layout for continuous catalytic gas-phase process for the manufacture of 1233zd (E) with hydrogen
fluoride isolation from HF/HCFO-1233zd mixture in a phase-separator. Recycling column (pos.314) is
introduced additionally for more efficient purification of the target product.
Fig.8 Layout for continuous catalytic gas-phase process for the manufacture of 1233zd (E) with hydrogen
fluoride isolation from HF/HCFO-1233zd mixture in a phase-separator. Recycling column (pos.314) is
introduced additionally for more efficient purification of the target product.
3. Liquid-phase process for the manufacture of trans-1-chloro-3,3,3-trifluoropropene (1233zd (Е)) by fluorination of 1,1,1,3,3-pentachloropropane (HCС–240fa) without catalyst [9–12]
Above it has been shown that 1233zd (Е) may be produced in catalytic liquid-phase processes at comparatively mild conditions with sufficiently high yields (see chapter 1). However, the presence of fluorinated catalysts favors the formation of «heavy» by-products, oligomers and resins which when accumulated in the reactor, dilute and deactivate catalyst. It results in loss of efficiency, and requires periodical outage and withdrawal of those products from the reactor. Those disadvantages might be debugged if fluorination of HCС–240fa is conducted without catalyst. In order to remove the limitations due to slow rate of such process, it has been proposed, keeping unchanged the general process layout for liquid-phase synthesis of 1233zd (Е), to conduct catalyst-less fluorination of HCС–240fa at more strict regime, under pressure of 150psig to 600psig (10,5–42atm) (preferably, at 350–450psig (25–32atm)) [9–12]. Various types of high-pressure reactors are applicable [11, 12]. It is optimal to combine them with one or more distillation column operated at lower pressure.
The process for the manufacture of 1233zd (Е) is periodical or continuous. Figure 9 illustrates a possible technological layout of continuous liquid-phase process for the manufacture of 1233zd (Е) without catalyst [11]. To soften the conditions and increase the process efficiency some researchers [10] offered a modification of continuous process for the manufacture of 1233zd (Е) using НСС–240fa fluorination in a number of reactors, where operational pressure is step-by-step decreased, and (correspondingly) step-by-step enrichment in target product occurs (see Figures 10 and 11). The required number of reactors depends on their size and desirable efficiency of the process. With larger size the result may be achieved with fewer reactors.
The published [9–12] data provide evidence for conclusion that under certain conditions HCС–240fa non-catalytic liquid-phase fluorination may be successfully realized in full-scale manufacture of 1233zd (Е) with purity at least 99,5%.
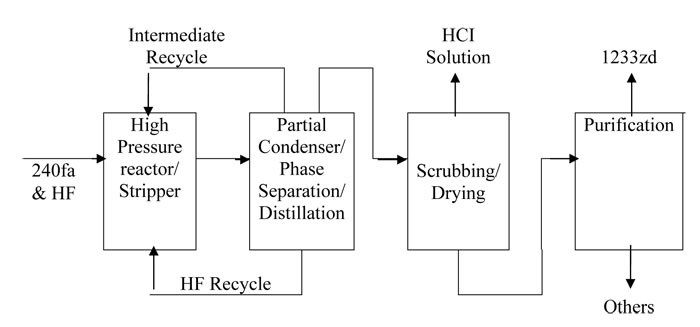
Fig.9 A variant of technological layout for continuous liquid-phase non-catalytic manufacture of 1233zd (Е).
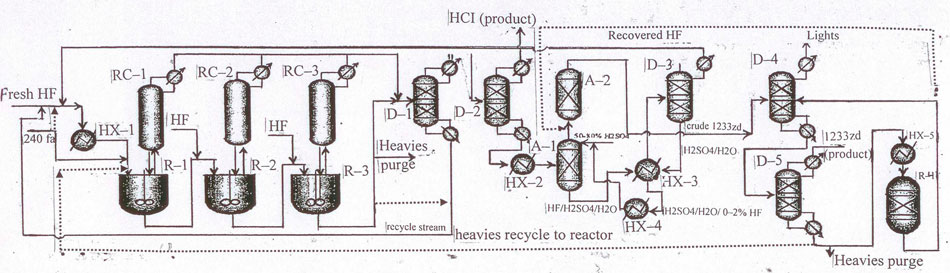 Fig.10 Layout for continuous liquid-phase process for the manufacture of 1233zd (E) with a sulfuric acid-based
variant of hydrogen fluoride isolation from HF/HCFO-1233zd mixture.
Fig.10 Layout for continuous liquid-phase process for the manufacture of 1233zd (E) with a sulfuric acid-based
variant of hydrogen fluoride isolation from HF/HCFO-1233zd mixture.
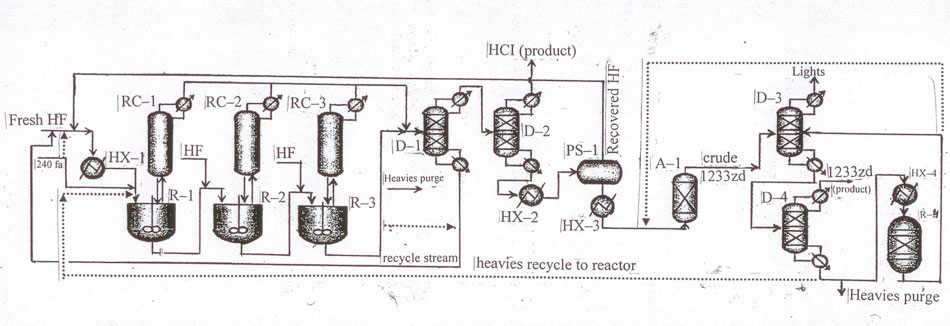 Fig.11 Layout
for continuous liquid-phase non-catalytic process for the manufacture of 1233zd (E) with hydrogen
fluoride isolation from HF/HCFO-1233zd mixture in a phase-separator.
Fig.11 Layout
for continuous liquid-phase non-catalytic process for the manufacture of 1233zd (E) with hydrogen
fluoride isolation from HF/HCFO-1233zd mixture in a phase-separator.
4. Manufacture of trans-1-chloro-3,3,3-trifluoropropene by catalytic dehydrohalogenation of 1,1,1-trifluoro-3,3-dichloropropane (HCFC–243fa) [13]or 1,1,1,3-tetrafluoro-3-chloropropane (HCFC–244fa)[14]
4.1 Manufacture of 1233zd (Е) based on 1,1,1-trifluoro-3,3-dichloropropane [13].
The process is based on catalytic dehydrochlorination of НСFС–243fa resulting in the formation of target product 1233zd (Е) and by-products (НCl, 1233zd (Z), etc.):
![]()
The process involves 4 main steps:
- Catalytic dehydrochlorination of НСFС-243fa generating the formation of a mixture of isomers 1233zd (Е+Z) and HCl;
- Isolation of HCl;
- Isolation and purification of target 1233zd (Е);
- Catalytic isomerization of 1233zd (Z) to 1233zd (Е).
Procedure 1: The process of НСFС–243fa catalytic dehydrochlorination is conducted in a gas-phase reactor (see chapter 2) using any available catalysts for dehydrochlorination (see chapter 2). Preferred catalysts are fluorinated metal oxides (Сr2O3, Al2O3), metal halogenides (CrF3, AlF3, AlCl3, FеCl3) and carbon-supported transient metals, such as Fe/C, Co/C, Ni/C, Pd/С. Original НСFС–243fa may be fed into the reactor (preferably via an evaporator) pure or mixed with some inert gas (nitrogen, argon etc.). Recommended temperature range is 300–450°С. The process is realizable in vacuum, at atmospheric pressure, or under increased pressure.
It is possible to regenerate catalyst periodically by any available method, e.g., passing air or air/nitrogen blend at 200–300°С during required time period.
Note 1: Optionally, dehydrochlorination of НСFС–243fa may be conducted through its treatment with some alkaline solution, e.g., KOH, NaOH, Ca(OH)2 etc., at increased temperature. The recommended temperature interval is between 50 and 70°С. The process is realizable in vacuum, at atmospheric pressure, or under increased pressure.
For procedures 2–4 scrubbing systems are applied, similar to those considered above for catalytic liquid-phase manufacture of 1233zd (Е) based on НСС–240fa.
Note 2: At least partially, cis-isomer 1233zd undergoes isomerization to trans–isomer at step 1 already, because dehydrohalogenation catalysts are very efficient in the process of 1233zd cis/trans isomerization.
4.2 The manufacture of 1233zd (Е) based on 1,1,1,3-tetrafluoro-3-chloropropane (HCFC–244fa) [14].
The process is based on HCFC–244fa gas-phase catalytic dehydrofluorination that results in the formation of 1233zd (Е) and some by-products (HF, 1233zd (Z), etc.):
![]()
The process involves 4 main steps:
- Catalytic dehydrofluorination of HCFC–244fa resulting in the formation of 1233zd (Е+Z) isomers mixture and HF;
- Isolation of HF;
- Isolation and purification of the target product;
- Catalytic isomerization of 1233zd (Z) to 1233zd (Е).
Procedure 1: Catalytic dehydrofluorination of HCFC–244fa is conducted in a gas-phase reactor (see chapter 2) under conditions that grant high conversion of HCFC–244fa and high selectivity of 1233zd (Е) formation. The preferred catalysts are metal oxides (Cr2O3), metal halogenides (FeCl3, FeCl3/C, AlF3, CeF4), and natural or synthetic graphite. Recommendable temperature ranges 300-350°С; contact time 10-60s.
In procedures 2-4 scrubbing systems are used similar to those used above for catalytic liquid-phase or gas-phase fluorination of HCC–240fa (see chapter 1, 2).
Conclusions
Among the considered processes for the synthesis of 1-chloro-3,3,3-trifluoropropene (catalytic liquid-phase hydrofluorination, catalytic gas-phase hydrofluorination, non-catalytic liquid-phase fluorination, catalytic dehydrohalogenation) the most purposeful for industrial implementation is non-catalytic liquid-phase hydrofluorination of 1,1,1,3,3-pentachloropropane in which case gumming in the reaction zone is minimized.
References
- US Patent 6844475 (Honeywell International Inc), 2005
- US Patent 6018084 (Daikin Industries Ltd.,), 2000
- US Patent Application 0201853 (Honeywell International Inc), 2013
- US Patent Application 0184786 A1 , 2012
- US Patent Application 0059200, 2011
- US Patent 8426656 (Honeywell International Inc), 2013
- US Patent Application 0172636 (Honeywell International Inc), 2011
- WO 066375 (ARKEMA FRANCE), 2012
- US Patent Application 0296127 Cottrell; Stephen A.; (Williamsville, NY) ; Tung; Hsueh Sung; (Getzville, NY), C07C 21/18, C07C021/18, 15.05.2012
- US Patent Application 0059199 Pokrovski K. A.; (Orchard Park, NY) ; Merkel D. C., C07C 17/20, C07C017/20, 29.08.2011
- US Patent Application 0211154 (Honeywell International Inc), C07C 17/23, C07C017/23, 04.02.2013
- WO 122860 (Honeywell International Inc), C07C 17/20, C07B 61/00, 22.08.2013
- US Patent 1829747 (Honeywell International Inc), C07C 17/23, C07C017/23, 09.07.2010
- US Patent Application 0271069 Wang Haiyou (Amherst, NY) ; Tung Hsueh Sung; (Getzville, NY) кл. C07C 21/18, C07C021/18, 20.04.2011
Recommended for publication by V. Kornilov
Fluorine Notes, 2015, 99, 3-4