Fluorine Notes, 2010, 73, 7-8
Fluorine notes, Vol. 6(73) 2010
PURIFICATION OF TETRAFLUOROETHYLENE USING THE REACTION MIXTURE COMPONENTS FOR SEPARATION AGENTS
V. S. Leont'ev
FSUE Russian Scientific Center "Applied Chemistry",
197198, Russia, St. Petersburg, Dobrolubov av. 14
Abstract:
The use of Difluorochloromethane as separating agent in the process of
rectification the Tetrafluoroethylene mixture with Trifluoroethylene was studied.
Keywords: tetrafluoroethylene, co-polymerization, perfluoro(3,6-dioxa-4-methyl-7-octene)sulfonylfluoride
Separation of tetrafluoroethylene - trifluoroethylene system by the method of rectification is one of the most complicated technological issues in tetrafluoroethylene purification.
In the blend to be separated trifluoroethylene is present in negligible quantity 0,033-0,034 %(wt). However, its content in the marketable product must not exceed 1 ppm. At atmospheric pressure the boiling point of tetrafluoroethylene is minus 76°C, and that of trifluoroethylene is minus 60°C. Tetrafluoroethylene - trifluoroethylene system is non-azeotrope, however, significant positive deviation from ideality are typical for it. At high tetrafluoroethylene concentrations the coefficient of relative volatility in tetrafluoroethylene - trifluoroethylene system is about 1,1.
At the same time difluorochloromethane is present in the reaction medium in amount of ~ 37,5%. The boiling point of difluorochloromethane is minus 40°C. It is easily separated from tetrafluoroethylene.
Rectification in a column with efficiency =63 theoretical plates, fed to the 11th plate from bottom, and reflux-to-product ratio R=17,5 results in the drop of trifluoroethylene content in the marketable product to less than 1 ppm [1].
Dichlorofluoromethane is not found in the marketable product (see Fig.1, Table 1). Heat load on the boiler is then 1,4 MJ/hour per 1 kg of the mixture under separation.
The tetrafluoroethylene - difluorochloromethane system exhibits larger positive deviations from ideality, then trifluoroethylene - difluorochloromethane system, thus providing good results when difluorochloromethane is applied for its separating agent. Introduction of this separating agent into the separated system does not contaminate it additionally. Considerable difference between b.p. of the products under separation, on one hand, and b.p. of the separating agent, on the other hand, makes power consumption at the stage of the agent regeneration incommensurable with that at the stage of tetrafluoroethylene from trifluoroethylene separation.
When the process is arranged with difluorochloromethane used for the separating agent (see Fig.2 and Table 2) and fed to the plate 46 (counted from the bottom upwards) in ratio 1,7:1 to the feeding then the content of trifluoroethylene in purified tetrafluoroethylene is also below 1 ppm. At the same time heat load on the column boiler is 0,64 MJ/hour per 1 kg of the mixture under separation.
Heat consumption for the regeneration of difluorochloromethane in C2 column is less than 0,1 MJ/hour per 1 kg of the mixture under separation.
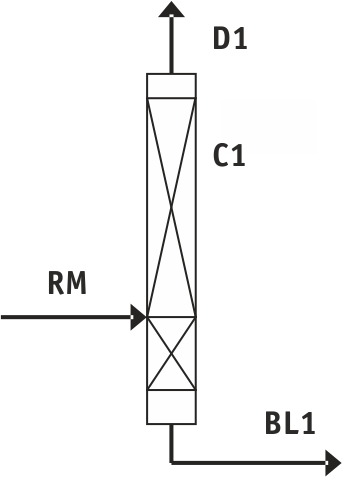
Fig.1. Purification scheme for tetrafluoroethylene without separating agent: C1 - rectification column
Table 1. Designation and composition of flows in Fig. 1
|
RM - mixture under separation: |
|
BL1 - bottom liquid of C1: |
|
D1 - purified tetrafluoroethylene |
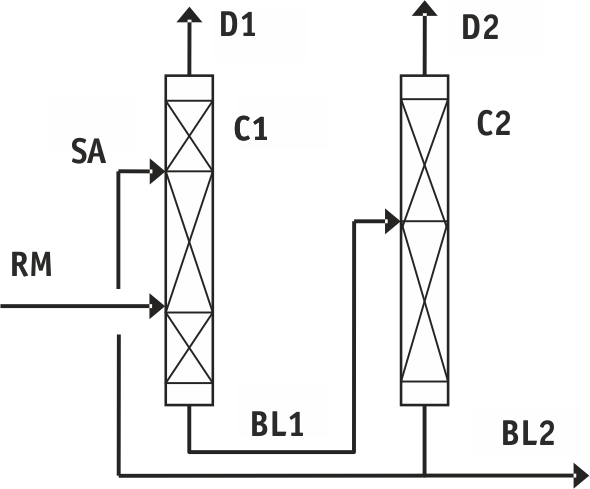
Fig.2. Flow diagram for the purification of tetrafluoroethylene with separating agent: C1 -tetrafluoroethylene purification column; C2 - difluorochloromethane regeneration column
Table 2. Designation and composition of flows in Fig.2
|
RM - mixture under separation: |
|
РA - separating agent (purified difluorochloromethane): |
|
BL1 - bottom liquid C1 (trifluoroethylene/difluorochloromethane mixture): |
|
BL2 - bottom liquid C2 (purified difluorochloromethane): |
|
D1 - purified tetrafluoroethylene |
|
D2 - trifluoroethylene fraction from industrial cycle: |
Therefore, the use of a component of the mixture under rectification (here: difluorochloromethane) for its separating agent, that is separated from the mixture and introduced into a specified zone of the rectification column, results in nearly 2 times decrease of power consumption in tetrafluoroethylene purification (taking into account the regeneration of difluorochloromethane).
References
1. V.S. Leont'ev, B. Nikiforov, chapter in book "Fluorine Compounds: Chemistry, Technology, Application"
, "Teza" publisher, S.-Petersburg, 2009, pp 343-351, ISBN 5-88851-06-8
Fluorine Notes, 2010, 73, 7-8
