Fluorine Notes, 2010, 73, 5-6
Fluorine notes, Vol. 6(73) 2010
Stage of initiation in the process of co-polymerization of tetrafluoroethylene with perfluoro-3,6-dioxa-4-methyl-7-octenesulfonylfluoride
O.S. Bazanova, A.S. Odinokov, L.F. Sokolov, V.G. Barabanov, B.N. Maximov, V.V. Kornilov
FSUE Russian Scientific Center "Applied Chemistry",
197198, Russia, St. Petersburg, Dobrolubov av. 14
Abstract:
The initiator concentration effects on the kinetics of solution radical
co-polymerization of tetrafluoroethylene with perfluoro(-3,6-dioxa-4-methyl-7-octene)sulfonylfluoride
are investigated
Keywords: tetrafluoroethylene, co-polymerization, perfluoro(3,6-dioxa-4-methyl-7-octene)sulfonylfluoride
Radical co-polymerization of tetrafluoroethylene (TFE) with perfluoro(3,6-dioxa-4-methyl-7-octene)sulfonylfluoride (FC-141) occurs according the following flow-diagram and results in the formation of F-4CF perfluorinated co-polymer:
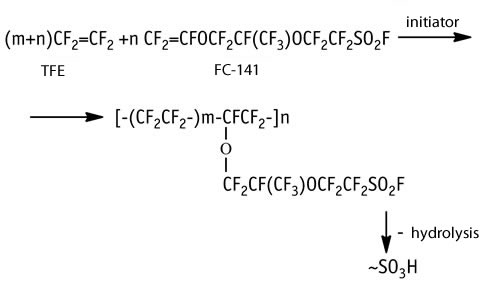
Sulfonylfluoride groups are hydrolyzed to sulfoacid groups. F-4CF is used in the production of proton-exchange membranes for fuel cells, for alkaline-chlorine electrolysis of sodium chloride, for hydrogen production via water electrolysis, and for heterogeneous acid catalyst [1-4].
In our earlier report [5] we considered the effects of the monomer mixture composition on the kinetics of TFE /FC-141 solution co-polymerization. This study investigates the initiator concentration effects on this process.
Experiment
Co-polymerization was conducted in a stainless steel (X18H10T) reactor of volume 200ml equipped with a jacket and a frame stirrer (n=300 rpm). The reactor was thermostated with the help of an ultrathermostat with accuracy ±0,1oC. 1,1,2-trifluoro-1,2,2-trichloroethane (freon-113) was used for solvent, initiator - bis(perfluorocyclohexanoyl)peroxide. Tetrafluoroethylene was cleaned from inhibitor (triethylamine) in an adsorber charged with activated carbon AG-3. The reactor pressure during the experiment (and hence TFE concentration in liquid) was kept unchanged by permanent TFE feeding from a calibrated buffer capacity. TFE consumption was determined from the pressure variation in the buffer capacity. When the depth of FC-141 conversion reached ~5% mass the polymerization process was stopped by triethylamine inhibitor introduced into the reactor. The resulting polymer was washed with chloroform and water and dried under vacuum at 60 oC to constant mass.
The copolymer composition was determined by the number of sulfofluoride groups as estimated by IR-spectroscopy [6], elemental analysis for sulfur [7], titration of sulfoacid groups of the hydrolyzed copolymer.
The co-polymer molecular mass was estimated by the maximal Newtonian viscosity (η0) of melt copolymer according to [8]. Those η0 values for the co-polymer samples were detected with the help of a capillary constant-pressure viscosimeter at various loads, the capillary diameter was 1,02mm, its length was 35mm, and temperature was 270oC.
The rate of co-polymerization was detected by gravimetric analysis by the co-polymer yield in polymerization.
It was found experimentally that the rate of TFE/FC-141 co-polymerization grows linearly when the initiator starting concentration increases within the range (7.5-47)*10-4 mol*l -1 (Fig.1), while the co-polymer equivalent weight (i.e., molecular mass per one sulfogroup) that is a characteristics of the co-polymer composition stays practically unchanged (Fig. 2).
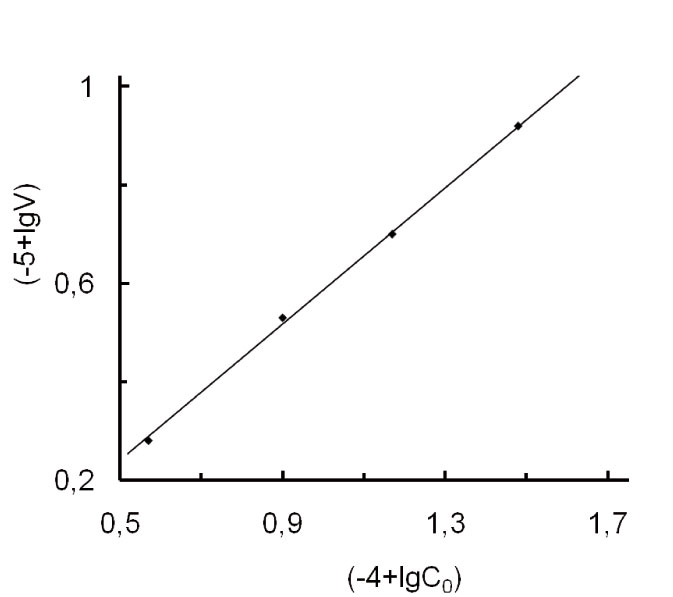
Fig. 1. Dependence of the rate of TFE/FC-141 co-polymerization (V, mol*(l*s)-1) on the initiator starting concentration (C0, mol*l-1) in logarithmical coordinates. The starting concentration of FC-141=1,2 mol*l-1. The concentration of TFE=1,04 mol*l-1.T=38±0,5oC, P=4,1atm
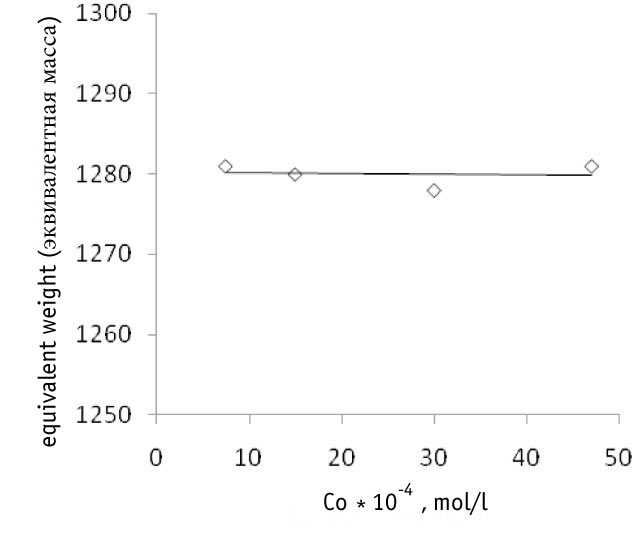
Fig.2. Dependence of F-4CF co-polymer equivalent weight on the starting concentration of the initiator (Co, mol*l-1) in the synthesis of co-polymer at 38oC, pressure 4,1atm, FC-141/TFE ratio =1,2/1,04 (mol)
According to the data of Fig.1 the co-polymerization reaction order (by the initiator) is n=0.7±0.1. The calculation was done by the Van't Hoff method [9, p.14] according to the formula:
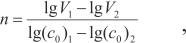
Here (co)1, (co)2, and V1, V2 are the starting initiator concentrations (mol*l-1) and related co-polymerization rates (mol*(l*s)-1) for points on the right line in Fig. 1.
The deviation of the reaction order from value 0.5 typical for quadratic chain termination to the side 1 associated with monomolecular chain termination is typical for polymerization followed by polymer precipitation from the solution [10]. The studied process belongs to that type of processes.
The viscosity of the studied F-4CF co-polymer samples melt at 270oC does not depend on their shearing force (fig. 3), i.e. the temperature of determination of melt flow index (MFI) was appropriately chosen, melt flow was Newtonian [11, p.190] and defined the co-polymer molecular mass.
The maximal Newtonian melt viscosity (η0) was obtained through the extrapolation of the melt flow curve (fig. 3) to the zero shearing force τ →0.
Therefore the data of Fig. 4 testify that when the starting initiator concentration increases not only the melt viscosity but also its molecular weight go down.
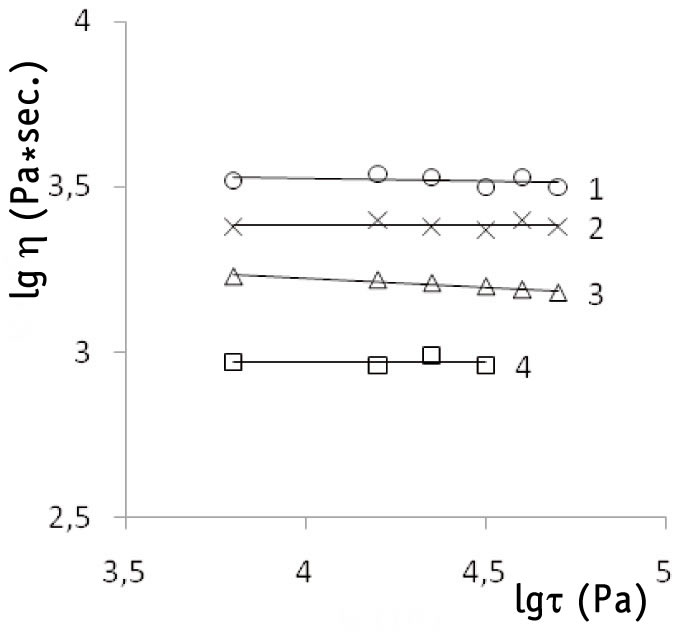
Fig. 3. Dependence of F-4CF co-polymer melt viscosity (η, Pa*sec) at 270oC
on the shear stress (τ, Pa) for polymer samples produced at various starting initiator concentrations
(Co).
1. Co=7,5*10-4 mol*l-1.
2. 15,0*10-4 mol*l-1.
3. 30,0*10-4 mol*l-1.
4. 47,0*10-4 mol*l-1.
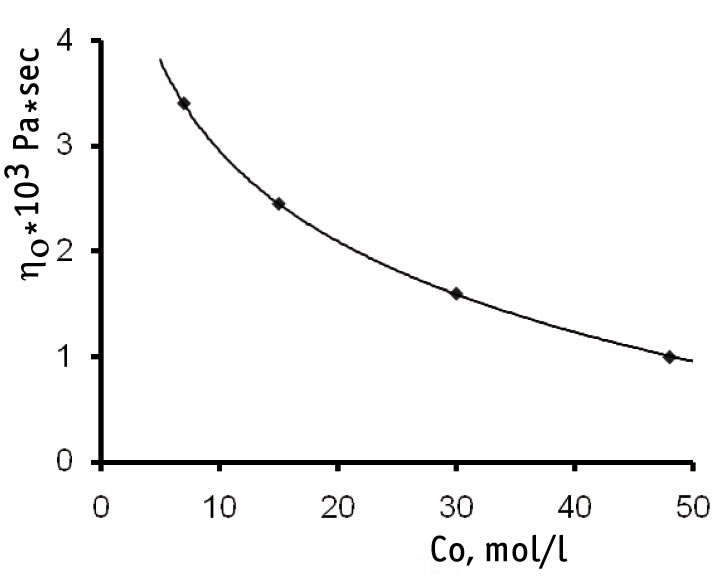
Fig. 4. Dependence of the maximal Newtonian viscosity (η0) of F-4CF melt co-polymer at 270oC on the starting initiator concentration (Co) in the process of F-4CF co-polymer synthesis.
Conclusions
1. In the process of radical solution TFE /FC-141 co-polymerization the increase of the initiator concentration
is accompanied by:
- linear growth of the co-polymerization rate,
- decrease of the molecular
weight of the produced co-polymer,
- unchanged composition (equivalent weight) of the co-polymer.
2. Order of the co-polymerization reaction by initiator is n=0.7±0.1.
The study was done within the framework of State Contract N 02.523.12.3022 financially supported by RF Federal Agency for Science and Innovations.
References
2. Yamabe M. Novoe v tekhnologii soedinenij ftora. M.: Mir, 1984. 521s.
3. Kirsh Yu. E., Smirnov S. A., Popkov Yu. M., Timashchev S. F.// Uspekhi khimii. 1990. t. 59. N 6. s. 970-994.
4. Waller F. G. // "Polimernye reagenty i katalizatory". M.: Khimiya, 1991. s. 46-63.
5. A. S. Odinokov, O. S. Bazanova, L. F. Sokolov, V. G. Barabanov, S. V. Timofeev.// ZhPKh, 2009. t. 82. N 1. s. 113-116.
6. Abramov S. P., Trofimova A. A., Barabanov V. G.// ZhPKh, 2009. t.82. N 12. s. 2043-2047.
7. Ryabnikova V. M., Nikolaeva A. P.// ZhAKh. 1976. t. 31. N 6. S. 1212-1214.
8. Marahonov I. A.// "Pererabotka plasticheskih mass". M.: Khimiya, 1966. s. 283.
9. Zahar'evskij M. S. Kinetika i kataliz. L.: LGU, 1963. 314 s.
10. Bagdasar'yan H. S. Teoriya radikal'noj polimerizacii. M.: Nauka, 1966. 300 s.
11. Vinogradov G. V., Malkin A. Ya. Reologiya polimerov. M.: Khimiya, 1977. 438 s.
Fluorine Notes, 2010, 73, 5-6
