Fluorine Notes, 2010, 73, 3-4
Fluorine notes, Vol. 6(73) 2010
Synthesis of perfluorodiacyl peroxides and kinetics of their thermal decomposition
O.S. Bazanova, L.F. Sokolov, B.N. Maximov
FSUE Russian Scientific Center "Applied Chemistry",
197198, Russia, St. Petersburg, Dobrolubov av. 14
Abstract:
Synthesis and kinetics of thermal decomposition are studied for a number of perfluorinated diacylperoxides applied for the initiators of co-polymerization of tetrafluoroethylene with perfluoro(-3,6-dioxa-4-methyl-7-octene)sulphonylfluoride
Keywords:Diacylperoxide, tetrafluoroethylene, co-polymerization
There are a number of published reports about the synthesis of perfluorodiacyl peroxides (PFDAPs) and their use for the initiators in the polymerization of fluorocarbon О±-olefins [1-5]. It is well-known that PFDAPs may initiate polymerization at relatively low temperature (~ 20-50oC), however, the kinetics of PFDAPs thermal decomposition is studied non-sufficient.
The present work reports about the synthesis and thermal decomposition of a number of available PFDAPs.
Synthesis of perfluorodiacyl peroxides
For feedstock in the synthesis of PFDAPs a number of fluoroanhydrides were used: perfluorocyclohexanoyl fluoride, dimers, and trimers of hexafluoropropylene oxides, and fluoroanhydrides formed as by-products in the synthesis of perfluoro(-3,6-dioxa-4-methyl-7-octene)sulphonylfluoride that is FC-141 monomer:
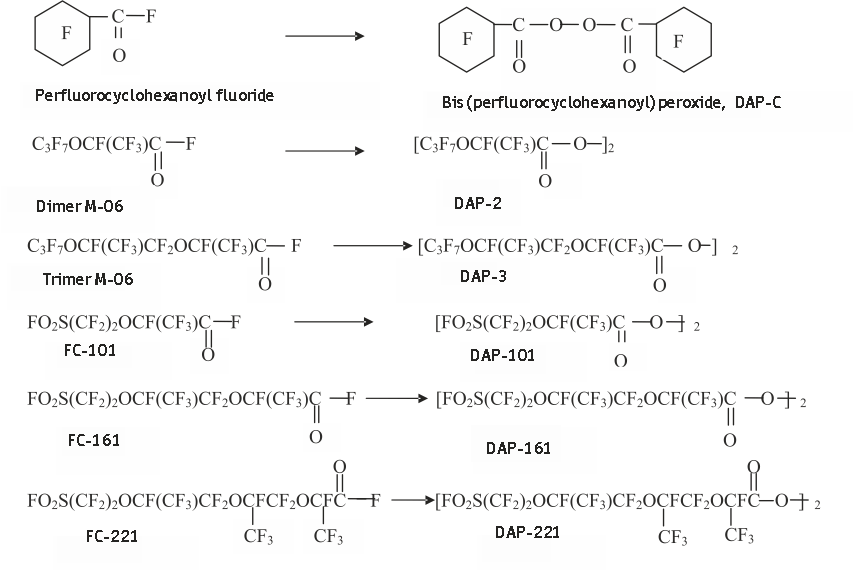
The results are shown in Table 1.
Table 1. Conditions for the synthesis of diacylperoxides (hereinafter: DAP)
Synthesized peroxide |
DAP-C |
DAP-2 |
DAP-3 |
DAP-101 |
DAP-161 |
DAP-221 |
||||||
Solvent |
R-113 |
FC-75 |
R-113 |
FC-75 |
R-113 |
FC-75 |
R-113 |
FC-75 |
R-113 |
FC-75 |
R-113 |
FC-75 |
| Concentration of peroxide, g/ml | 0,062 |
0,068 |
0,045 |
0,047 |
0,040 |
0,038 |
0,009 |
0,007 |
0,015 |
0,010 |
0,021 |
0,012 |
Yield, % mass |
78 |
86 |
56 |
59 |
47 |
45 |
30 |
25 |
38 |
26 |
72 |
39 |
Both 1,1,2-trifluoro-2,2,1-trichloroethane (R-113) and perfluoro(2-butyltetrahydrofuran) (FC-75) were used for solvents.
Using DAP-C for an example we studied the impact of some factors, such as temperature, rate of stirring and various solvents on the yield of the product (Table 2).
Table 2. The effect of the synthesis conditions on the yield of PFDAP
В |
Temperature,oC (n=760 rpm, R-113) |
Rate of stirring n, rpm (T= -10oC, R-113) |
Solvent (T=-10oC, n=760 rpm) |
||||||||||
-10 |
0 |
10 |
18 |
100 |
300 |
500 |
760 |
R-113 | FC-75 | MD-46 | RC-316 | CHCl3 | |
| Concentration, g/ml | 0,062 |
0,048 |
0,036 |
0,027 |
0,042 |
0,049 |
0,054 |
0,06 |
0,062 |
0,068 |
0,038 |
0,04 |
0,022 |
Yield, % |
78 |
60 |
45 |
34 |
53 |
62 |
68 |
75 |
78 |
86 |
48 |
52 |
28 |
The applied solvents:
1,1,2-trifluoro-2,2,1-trichloroethane (R-113); perfluoro(-2-butyltetrahydrofuran) (FC-75); perfluoromethyldiethylamine (MD-46); hexafluorodichlorocyclobutane (RC-316); chloroform (CHCl3).
Effect of temperature of synthesis: when temperature rises from -10 to +18oC (see Table 2) the product yield drops. Apparently, the number of by-reactions and the yield of by-products grow with the temperature of synthesis.
Effect of rate of stirring: the yield of peroxide grows with the rate of stirring (from 100 to 760 rpm (see table 2). The reason is that intensive stirring improves the contact between reagents from different layers (water and organic) of the reaction mass.
Effect of solvent: from the data of Table 2 one may conclude that different solvents provide different effects on the yield of peroxide. It may be connected to the difference in solvent polarities. The higher is the solvent polarity, the higher is the product yield, because the electrostatic forces between the solvent particles and solute molecules grow thus accelerating the reaction and increasing the finish product yield.
Decomposition of perfluorodiacylperoxides
Thermal decomposition of peroxides was conducted in freon R-113 environment in a tightly closed glass test-tube placed into an ultra-thermostat. Its temperature was kept with accuracy
В±0,1oC. In equal patches the peroxide concentration was detected by iodometric titration, the graph of its dependence on the decomposition period was constructed, and the half-life time of peroxide was determined. Te results are shown in Table 3.
Table 3. Thermal decomposition of PFDAPs
half-life
|
peroxide/ ToC |
DAP-C |
DAP-2 |
DAP-3 |
DAP-101 |
DAP-161 |
DAP-221 |
30 |
49 h |
1 h |
2 h 30 min |
2 h 57 min |
3 h 6 min |
3 h 56 min |
40 |
13 h 45 min |
27 min |
46 min |
1 h 55 min |
2 h 7 min |
2 h 45 min |
50 |
1 h 18 min |
В |
В |
В |
В |
В |
The data of Table 3 evidence strong dependence of the PFDAPs thermal decomposition rate on the structure of its acyl-group, making it possible to choose the optimal PFDAP type in line with the designed polymerization process temperature.
PFDAPs thermal decomposition is a complicated process that involves both homolysis by one or more bonds and non-radical mono-molecular decomposition via cycle transient state, whereas those two are difficult to separate kinetically. The mechanism study is of large applied significance for improvement of traditional ways of unsaturated substances polymerization [7], that make use of PFDAP for the process initiators. The results of kinetic treatment of experimental data on PFDAP thermal decomposition are presented in Table 4:
Table 4.Thermal decomposition of PFDAPs (R-113 solvent)
Peroxide |
Temperature interval,oC |
Decomposition rate constant, s-1 |
DAP-C |
40-60 |
7,9*1018exp(-22200/RT) |
DAP-2 |
30-50 |
3,8*1014exp(-23100/RT) |
DAP-3 |
30-50 |
2,9*1014exp(-22700/RT) |
DAP-101 |
30-50 |
2,5*1014exp(-21800/RT) |
DAP-161 |
30-50 |
2,6*1012exp(-25000/RT) |
DAP-221 |
30-50 |
1,9*1010exp(-26200/RT) |
The data from Table 4 testify that PFDAPs thermal decomposition as to compare with that of non-fluorinated analogues requires much lower activation energy, whereas the elongation of PFDAPs perfluorinated radical results in some decrease of its thermal decomposition activation.
Experiment
PFDAP synthesis is based on the interaction between perfluorinated carboxylic acid haloidanhydride with aqueous solution of sodium peroxide formed through the interaction of sodium hydroxide with hydrogen peroxide. The synthesis is conducted in a glass round flask of volume 500 ml equipped with a stirrer, drop-funnel and thermometer placed into a bath at minus 10 - minus 15oC (ice/sodium chloride blend). At minus 15oC under stirring (n = 300 rpm) the flask is charged through the funnel with as follows: freon R113, saturated aqueous solution of NaCl, 30% aqueous solution of NaOH, and 30% solution of H2O2.
The flask content is kept till the reaction medium temperature reaches minus 8 - minus 10oC, and fluoroanhydride of perfluorocarboxylic acid from a drop-funnel is added dropwise under continuous stirring into the flask at such rate that the reaction mixture temperature would not exceed minus 7oC.
As all fluoroanhydride is added the flask with reaction medium is kept at minus 7 - minus 10oC under stirring for 30 minutes. Then saturated aqueous solution of NaHCO3 cooled to minus 8oC is added into the flask from a drop-funnel for neutralization, and kept for 15 minutes under stirring.
The flask matter is then moved into a drop-funnel, kept for 5 minutes, its bottom fluorocarbon layer including perfluorodiacylperoxide solution is then separated, triply washed in a separation funnel with saturated NaCl solution cooled to minus 10oC, its fluorocarbon layer is then separated, put into a polyethylene jar, sealed and kept in a freezer at -10oC or lower.
The in-solvent peroxide concentration is detected by iodometric titration according to the technique that uses acetic acid and chloroform [6]. A mixed solvent is applied in order to increase the solvency of acetic acid. The flask is charged with a sample containing 0,1 gram-equivalent of peroxide, 20 ml of solvent (acetic acid : chloroform = 3:2) is added, and the flask is blown with inert gas. After adding of 1ml of fresh saturated solution of sodium iodide the flask is put in a dark place, then the flask content is titrated with 0,1N solution of sodium thiosulfate till vanishing of yellow color. Underestimation may happen if the solvent contains more chloroform than acetic acid, because ionizing solvent is necessary, and the quantity of chloroform should not exceed that needed for the sample dissolving.
The calculation of perfluorodiacylperoxide concentration is done according to formula as follows:
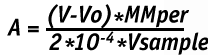
Here:
V is quantity of sodium thiosulfate spent in titration, ml
Vo is quantity of sodium thiosulfate in blank experiment, ml
MMper is molecular mass of the synthesized peroxide, g/mole
Vsample is the sample volume, ml.
Conclusion
Therefore, the study resulted in development of a method for the manufacture of perfluorodiacylperoxide intended for the process of fluoromonomer polymerization starting with available В fluoroanhydrides of perfluoroacids of various structure.
The study was done within the framework of State Contract N 02.523.12.3022 financially supported by RF Federal Agency for Science and Innovations.
References
1. US Patent 2559630, 1951
2. US Patent 2580358, 1951
3. US Patent 3642742, 1972
4. US Patent 3674758, 1972
5. D.E. Ryce, C.L. Sondberg, Polymer Preprints, 12, 276, 1971.
6. V.L. Antonovsky, M.M. Buzlanova "Chemical and chromatographic methods of organic peroxides analysi. Reviews of selected chemical industry"- NIITECHIM, Moscow, 1972, Issue 46, p. 5
7. Marco R., Shreeve J.M. - Adv. Inorg. Chem. A Rad., 1974. N 16, СЂ.110-169.
Fluorine Notes, 2010, 73, 3-4
