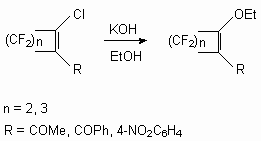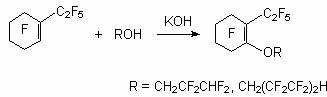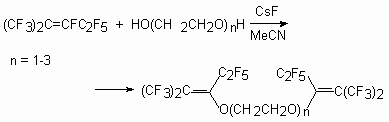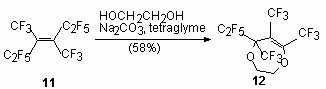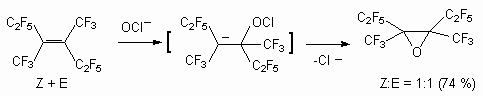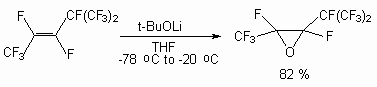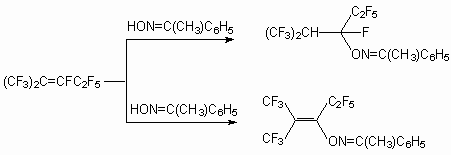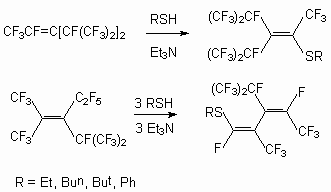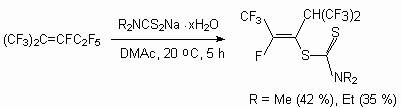Fluorine Notes, 2000, 12, 1-2
INTERNAL PERFLUOROOLEFINS IN A SYNTHESIS OF FLUOROORGANIC COMPOUNDSG.G. FURIN Institute of Organic Chemistry, 630090, Novosibirsk, Russia
CONTENTS 1. Introduction 2. Synthesis of partially fluorinated organic compounds using the reaction of internal perfluoroolefins with nucleophilic reagents.
3. Synthesis of perfluorinated compounds internal perfluoro-olefins and elementary fluorine.
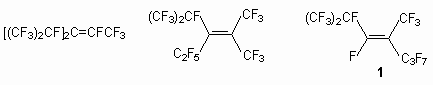
4. Conclusion References. Recently, considerable attention has been given to the development of ways to obtain new fluorine-containing compounds usually with various functional groups. Accessible perfluoroolefins are widely used for or a synthesis of some fluorine derivatives, while for others we use oligomers, whose distinctive ability is brightly expressed in electrophilicity [1]. These properties perfluoroolefins -new make a basis of one of directions of fluoroorganic chemistry including study of reactions them with nucleophilics by reagents, which has appeared olefinic systems as in the theoretical plan, as allows deeply to penetrate into properties perfluoroolefins of system, and in practical aspect doing accessible many fluoroorganic compounds. Due to specific influence of atoms of a fluorine on the not saturated system of reaction with nucleophilics by-reagents not only supplement chemistry of olefins, but also show their unique properties. So, among these compounds the substances with interesting electrophysical and thermophysical properties, with high physiological activity are revealed, which creates the base for a synthesis of medicinal preparations, lubricant materials, liquid dielectrics and heat-carriers, of highly effective surface- active substances and many materials for other spheres of the appendices [2-8]. The regular research of reactions ramified linear and cyclic perfluoroolefins basically mentions three aspects of reactions most typical for a number of internal perfluoroolefins, by virtue of specific influence of atoms of a fluorine on olefins system: 1) reactions with nucleophilics by-reagents, 2) reaction with radicals and oxidizings agent; 3) reaction with electrophilic reagents including processes, proceeding in superstrong acids and in SbF5environment. Highly fluorinated alkenes are generally very electron-deficient species and therefore susceptible to attack by a wide range of nucleophiles. Reactions between perfluoroolefins and nucleophilic by- reagents are key processes in chemistry of fluoroorganic compounds. Both are being widely studied and are used in synthesis of various derivatives including heterocyclic compounds [9-11]. Of special interest are reactions between internal new perfluoroolefins and nucleophilic by-reagents, as opposed to terminal perfluoroolefins (basically perfluoropropylene, perfluoroisobutylene, tetrafluoroethylene). The influence of perfluoroalkyl groups and fluorine atoms towards the olefin bond creates the preconditions of realization of a number of unusual processes of addition - elimination, being characteristic for the occurring of reactions with nucleophilic reagents, and allowing to obtain the variety of products. The wide range of nucleophilic reagents in reactions with compounds containing an electronic-saturated double bond allow to obtain products of connection on this communication, and to synthesize various heterocyclic compounds with perfluoroalkyl groups. In a number of reviews the authors mention the reactions between perfluoroolefins and nucleophilics by-reagents [12-15]. However, the basic reaction with C -nucleophiles and with binucleophilic reagents results in a synthesis of heterocyclic compounds, whose aspects are not examined in the present review. At the same time, one of most important problem is the problem both the development of approaches to synthesis of partially fluorinated compounds on basis of internal perfluoroolefins and of the opportunity to form on their basis the active intermediate particles (anions, radicals, carbocations) and to involve them in the reaction with organic substrates [12,16]. By virtue of it the basic attention in the review is given to the analysis of the saved experimental material on: 1) reactions of internal perfluoroolefins with nucleophilic by-reagents, 2) research of obtaining generation anion and radical particles containing perfluorinatered fragment, which can be used in quality sinthons, 3) revealing olefinic features of the system having perfluoroalkyl groups as substituents. The present review is particularly focused on the above-mentioned chemical transformations and reactions with various reagents. Taking into account, that alcohols and the amines can make a basis for obtaining a number of completely fluorinated organic compounds for different practical purpose, we have considered it worthwhile to analyze the data on their reactions with the purpose of formulating the basic laws of chemical processes and in order to emphasize the particular problems of interest for the modern internal perfluoroolefins chemistry and the known ways of their solution Reactions of terminal perfluoroolefins with nucleophilic reagents have been studied in detail and play an important role in a synthesis of key intermediate products of organic synthesis. However, insufficient attention has been given to reactions of internal perfluoroolefins with nucleophilic reagents. At the same time, internal perfluoroolefins are of interest by themselves as they can be considered raw material for a synthesis of new organic compounds [1-8]. The assumption is based on the data of photoelectronic spectroscopy of the perfluoroalkyl group at double bond lower energy p-orbital, which promotes attack of nucleophile on the atom of carbon of this bond. The potentials of a reduction are the important characteristic of the olefinic system reactionary ability [14]. The size depends on the nature of the substituent at double bond and upon the transition from the substituent fluorine to polyfluoroalkyl, it is decreased. With the increase of the number of perfluoroalkylic groups at double bond, the potential of reduction is lowered, and the reactionary ability perfluoroolefins grows: (RF)2C=C(R2)2 > (RF)2C=CFRF> (RF)2C=CF2 , RFCF=CFRF> RFCF=CF2 > CF2=CF2. The fluorine polyfluoroalkyl group essentially stabilizes an intermediate carbanion [17-19]. All this causes the reactions between internal perfluoroolefins and nucleophilic by-reagents to have a decrease in reactivity in the series olefins and we can see that it corresponds to a decrease in the stability of the derived intermediate carbanions. However, in these reactions it is necessary to take into account the influence of the structure of internal perfluoroolefins. Factors influencing electron-affinity of the fluorinated alkenes, i.e. the influence of fluorine or perfluoroalkyl (RF) as a substituent at the double bond, are important for consideration, as are the effects of these substituents on intermediate carbanion stability. This is insufficient to account for the greater reactivity of perfluoropropene, as compared to perfluoro-2-butene because the corresponding intermediate {Nu-CF(CF3)-CFCF3}- can only have the stability marginally different from that of [NuCF2CFCF3]-. Perfluoropropylene oligomerixes readily produce a mixture of dimers and trimers in the presence of a base or fluorine ion. At the same time nucleophilic reactions of the dimers and trimers have been investigated by several researches. As to trimers, three isomers are known at present : perfluoro-3(1-methylethyl)-4-methylpent-2-ene, perfluoro-3-ethyl-2,4-dimethylpent-2-ene and perfluoro-2,4-domethylhept-3-ene (1).
2.1. The interaction of internal perfluoroolefins with O-nucleophilic reagents in the synthesis of fluoro-organic materials and intermediate products. The interaction of internal perfluoroolefins with alcohols proceeds only
in the presence of the bases, leading to the products of the addition
and the substitution of the fluorine atoms, as well as the products of
more complex transformations [20]. Interaction of polyfluorinated alcohols
with metal sodium in excess of alcohol gives corresponding sodium alcoholates
[21-23]. However, usually the polyfluorination reaction of perfluoroolefins
and polyfluoroaromatic compounds proceeds without preliminary allocation
of alcoholates of alkaline metals, but at the presence of the bases.
For example, NaOH [24,25], triethylamine, Hg(OAc)2 [26] ,
etc have been used. The interaction between polyfluorinated alcohols
and perfluoroolefins in the presence of sodium hydride yields [26] the
products of fluorine atom substitution at double bond. CsF has proved
an effective catalyst also in reactions of perfluoroolefins with
The route of reaction for internal perfluoroolefin is determined substantially by the nature of substituents in aryl fragment, providing the possibility of synthesis of compounds with required structure by variation of these substituents. The relative smoothness of the course of connection and replacement reactions under the influence of a nucleophilic reagent on a perfluoroolefin is determined by spatial and electronic effects in a C-carbanion, forming as an intermediate product. The direction of the primary attack of the alcocsilic anion also is determined by the electronic and spatial factors. Thus, the action of methylic alcohol and phenol on trans-1,1-dichlorohexafluorobut-2-ene gives products of addition by carbon atom of double bond in a position 2 [28].
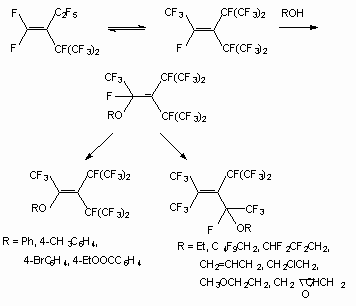
The double bond in perfluoro-2-methylbut-2-ene is extremely susceptible to nucleophilic attack and even reacted with neutral methanol (20 oC, 6 h) to produce 2-methoxy-nonafluoro-3-methylbutane and 2-methoxy-nonafluoro-3-methylbut-2-ene [29].
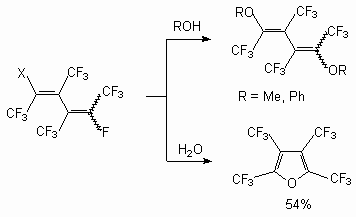
As shown in [30], the interaction between methanol and dimers of hexafluoropropylene in the presence of a KOH or triethylamine in a tetrahydrofuran (acetone, acetonitrile, N,N-dimethylformamide (DMF)) leads to the products of a fluorine atom substitution at double bond as well as the products of addition to it. Depending on ratio of reacting molecules formation of methylic ethers of the non-saturated perfluorinated acids and substituted ketones has observed [20]. Simultaneously, there is also a process of two methoxy groups entry in perfluoro-2-methylpent-2-ene, which produces isomeric 1,3-dimethoxyperfluoro-2-methylpent-1-ene (yield 51 %). Perfluoro-4-methyl-pent-2-ene reacts only with a sodium methylate, producing isomeric 1,3,4-trimethoxy-perfluoro-2-methylpent-1-ene (ratio 3:1) (yield 57 %) [31]. Perfluoro-2-methylpent-2-ene reacts with a,a,w-trihydroperfluoroalcohols with formation of the products of fluorine atom replacement at double bond [27]. It is possible to use t triethylamine and pyridine as the catalyst instead of CsF. The dimer of perfluorobut-2-ene reacts not only with methanol and phenol, yielding disubstituted products, but also with water, yielding perfluorotetramethylfuran [32].
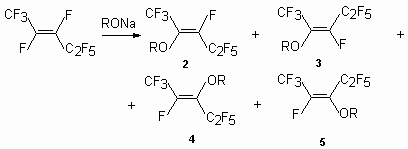
Sodium methylate and sodium ethylate in the reaction with trans-perfluoropent-2-ene form a mixture isomers (2), (3), (4) at the ratio of 9:1:4.5 (general yield 60.2 %), whereas the influence potassium phenolate in suspension of tetrahydrofuran at 0-10 oC results in a mixture of isomers (2) - (4) and isomer (5) [33].
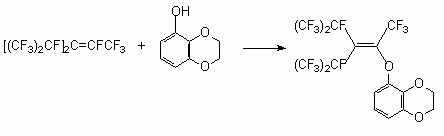
Phenols in the presence of Et3N with a number of internal perfluoroolefins allow to obtain exclusively the products of a fluorine atom replacement at double bond. Thus, the action of para-cresole on perfluoro-2-methylpent-2-ene [34], 3-oxybenzdioxane on hexafluoropropylene trimer in the presence of Et3N [35], and also the action of methyl ester 4-oxybenzene acid on tetrafluoroethylene pentamer in the presence of NaOH [36] yield the products of fluorine atom substitution at internal double bond.
It is used for a synthesis of various surface-active substances.
4-HOC6H4SO3Na reacted with perfluorononene in the presence of Et3N in DMF at 40 oC for 2 h to produce potassium salt [(perfluorononenyl)oxy]benzenesulfonate [37]. The similar results are received for reactions of a phenol with dimers and trimer of a hexa-fluoropropylene in the presence of Et3N [38-42].
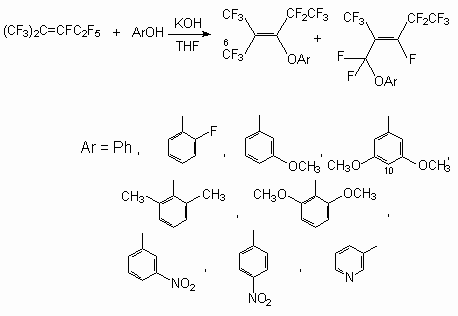
Some nucleophilic reactions of trimers 1 in particular with phenol have been reported by some authors [40-42]. Sodium phenoxide readily reacted with trimer 1 in various solvents at room temperature. Thus, phenyl-perfluoro-2,4-dimethylhept-4-ene-3-yl and perfluoro-2,4-dimethyl-hept-3-ene-3-yl ethers were obtained as major products, together with a small amount of phenyl-perfluoro-2,4-dimethylhept-3-ene-5-yl ether [41]. In apropos polar solvents such as DMF, DMSO or MeCN, the reactions gave mixture of di- and tri-phenoxylated products even when only one equimolar amount of sodium phenoxide was used. In less polar solvent, such as diglyme, THF, Et2O, the reaction proceeded more moderately, and the monophenoxylated product was obtained. When sodium phenoxide was allowed to react with perfluoroolefin 1 in the presence of Et3N, the product was a mixture of 6 and 7 in ratio of 2:1.
In case of isomers mixture of a hexafluoropropylene trimers the reaction
with alcohols and replaced phenol results two various products [43].
The primary act of these reactions is the attack of O-nucleophilic reagent
on the atom of carbon at a double bond having atom of a fluorine. The
further transformations of C-carbanion depend on the nature of O-nucleophile:
in reactions with alcohols the fluoride-ion is eliminated from
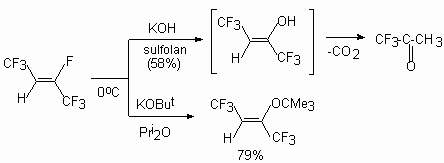
At the same time authors of the [43] have shown a possibility of izomerization of allylic ethers to vinylic ethers under influence of a cesium fluoride. It is important to understand clearly the necessity of strict adherence to the conditions for the occurring of such processes involving nucleophilic reagents and the relative role of substrates in this reaction. (E) and (Z) -2-tert-butoxyperfluorobut-2-enes are formed [44] under the influence of potasium tert-butylate in triglyme at 0 oC on a mixture of E and Z-perfluorobut-2-enes (4:1). 2H-Heptafluorobut-2-ene under action of the bases does not eliminate the hydrogen fluoride with formation of acethylene bond; only the substitution of fluorine atom at a double bond. Thus, the action of KOH in sulpholane on this compound leads to 1,1,1-trifluoroacetone, and the action of potassium tert-butylate in diisopropylic ether yield the product of substitution of fluorine atom at double bond compound (8) [45].
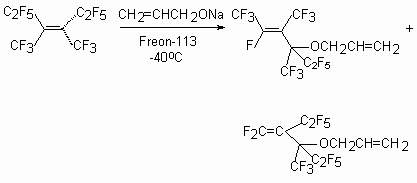
The reaction of tetramer tetrafluoroethylene with sodium derivative of allylic alcohol in Freon-113 at -40 oC leads to two products [46].
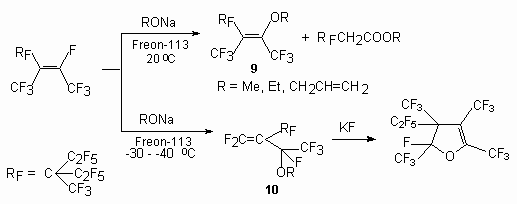
The pentamer of tetrafluoroethylene contains rather mobile atom of a fluorine at internal double bond and at action is alcoholic in the presence Et3N or sodium alcoholate. The structure of the products depends on temperature of the process [47,48]. When the pentamer tetrafluoro-ethylene reacts with alkoxide anions at low temperature (-30 to -40 oC), the product 10, corresponding to the reaction route by kinetic control is isolated, whereas at higher temperatures, thermodynamic control prevails to give the products 9. Compound 10 was converted to dihydrofuran by heating with KF in sulfolane [47].
The similar situation is observed and in the reaction of perfluorocycloolefins with alcohols [20,33]. In the reaction of 2-chloroperfluorocycloalkenes with KOH in ethylic alcohol the products of chlorine replacement on ethoxy-group are formed [49].
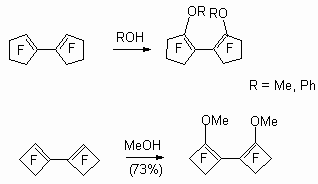
Perfluorobicyclopentene and perfluorobicyclobutene react with methylic alcohol and phenol, yielding products of replacement of a fluorine atoms at double bond, that is, disubstituted methoxylic or phenoxylic derivative [32].
As it was shown earlier, phenol reacts with perfluoro-2-methylpent-2-ene in the presence of a KOH in tetrahydrofuran and produces [3,3,3-trifluoro-1-pentafluoroethyl-2-trifluoro-methyl-propenyloxy]benzene with replacements of fluorine atom at internal double bond [50].
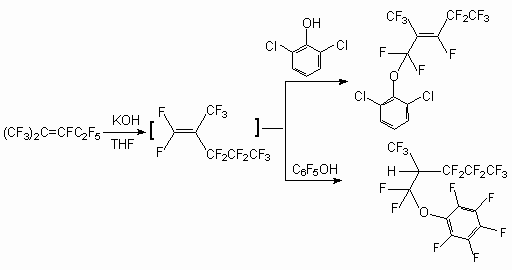
It was shown by us, that the entry into reaction with perfluoro-2-methylpent-2-ene phenols of electronodonating substituents (2-fluorophenol, 3-methoxyphenol, 3,5-dimethoxyphenol) does not change character of formed products. At the same time realization of the reaction under the same conditions with 2,6-dichlorophenol and pentafluorophenol produces (1,1,3,4,4,5,5,5-octafluoro-2-trifluoromethyl-pent-2-enyloxy)-2`,6`-dichlorobenzene and (1,1,3,3,4,4,5,5,5-nonafluoro-2-tri-fluoromethylpentyloxy)-pentafluorobenzene respectively [50].
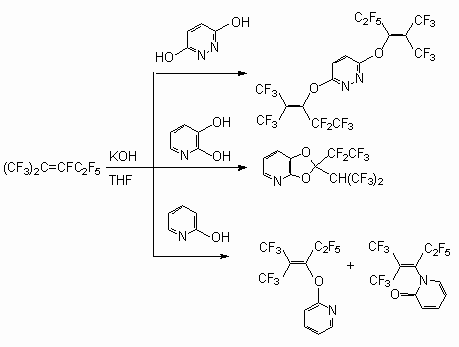
As available in a molecule of two OH-group the reaction proceeds with participation of both nucleophilics of the centres. Thus, the perfluoro-2-methylpent-2-ene with pyradazine-3,6-diol yields the 3,5-bis[3,3,3-trifluoro-1-pentafluoroethyl-2-trifluoromethyl-propenyloxy]pyridazine, and with 2,3-dioxypyridine - 2-pentafluoroethyl-2(2,2,2-trifluoro-1-trifluoromethyl-ethyl)-[1,3]-dioxolo[4,5-b]pyridine [50].
In the reaction of perfluoro-2-methylpent-2-ene with 2-oxypyridine two products are formed, that is, 2[3,3,3-trifluoro-1-pentafluoroethyl-2-trifluoromethyl-propenyloxy]pyridine and 1[3,3,3-trifluoro-1-pentafluoroethyl-2-trifluoromethyl-propenyl]-1H-pyridin-2-one respectively, which is caused by a fluorine atom replacement at double bond under influence O- and N- nucleophiles [50]. The interaction of perfluorocyclobutene with KOCH2Ph issue a product of fluorine atom replacement at double bond with high yield [51]. The behavior of polyfluorinated alcoxylate is similar with alcohols [27].
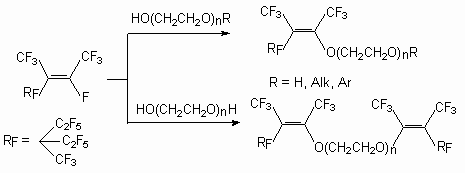
One of the important factors influencing on internal perfluoroolefins reactionary ability is the compounds structure [52]. It can be seen, for example, in reactions of methylic alcohol with the following three compounds: decafluorocyclobuthyl-1,1’-diene effectively reacts with neutral methylic alcohol, tetradecafluorobicyclopentyl-1,1'-diene reacts with it within several days, whereas reactions with octafluoro-3,4-bis(trifluoromethyl)-hexa-2,4-diene need the presence of the basis [52]. This is the illustration of the structural factors influence, because the electronic factors are the same for every compounds. Tetramer, pentamer and hexamer of tetrafluoroethylene during reaction with ethyleneglycols issue the products of fluorine atom replacement at double bond, which are successfully used as effective superficially active substances [53,54].
As it has been shown [55], during the reaction of perfluoro-2-methylpent-2-ene with mono-, di-, and triethylene-glycols at a reagents ratio 3:1 in MeCN (or THF, DMF, acetone) in the presence of Et3N (or KOH), the following partially fluorinated vinilic diethers are formed.
At the same time the main direction of the reactions between internal perfluoroolefins and ethylene-glycol is the formation of cyclic compounds - derivative 1,3-dioxolane. These processes are described for following reactions - ethyleneglycol with perfluorobut-2-ene [56], perfluoro-2-methylpent-2-ene [57, 58], pentamer tetrafluoroethylene [47,59,60], perfluoro-3,4-dimethylhex-2-ene [61]. For example, the interaction of equimolar amounts of ethyleneglycol with perfluoro-2-methylpent-2-ene in bipolar aprotic solvents in the presence of a triethylamine yields a mixture of products containing 2(pentafluoroethyl)-2(2,2,2-trifluoro-1-trifluoromethyl-ethyl)-1,3-dioxolane (72-81 %), 5,7-difluoro-5(pentafluoroethyl)-6-trifluoromethyl-3,5-dihydro-2H-1,3-dioxypine (3-5 %) and (5,5-difluoro-7(pentafluoroethyl)-6-trifluoromethyl-3,5-dihydro-2H-1,3-dioxypine (19-28 %)) [58,59]. At the reaction perfluoro-3,4-dimethylhex-3-ene (11) with ethylene-glycol 5-pentafluoroethyl-5.6.7-tris-(trifluoromethyl)-1,4-dioxypine (12) is obtained [61].
The isomerization is not found in the products of reaction between perfluoro-4-methylpent-2-ene and ethylene-glycol in a tetrahydrofuran (with KOH presence), and in the given conditions the derivative of a 1,4-dioxane (13) is formed with a low yield [55].
Reactions of internal perfluoroolefins with sodium hypochlorite in alkaline medium (at the presence of acetonitrile) is a sample of epoxidation, including the addition of OCl-anion to double bond and further substitution of chlorine atom by intramolecular attack by intermediate C-carbanion [62-67].
Lithium tert-butylperoxide has similar properties. It has been used effectively as an epoxidising agent with electron deficient alkenes [68,69].
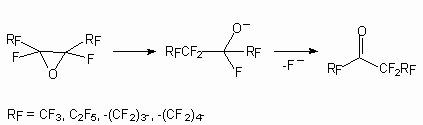
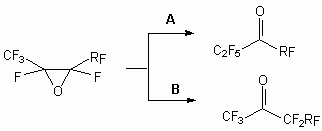
The application of this methodology to fluorine containing systems is essentially enriched by our knowledge of perfluoroolefinic system properties. The reagent’s nature is important too. Thus, calcium hypochlorite was ineffective for epoxidation of fluorine-containing olefins sometimes, whereas lithium tert-bulylperoxide system was extremely effective [70,71]. The activity of perfluoroolefins in epoxydation reactions is corresponding to the activity of nucleophilic reagents. That fact, that oxides of perfluoroolefins easily react with fluoride ion, has allowed to develop a convenient method of synthesis of perfluorinated ketones. It is known perfluoroolefin oxydes easily rearrange, that have allowed to develop a convenient method of perfluorinated ketones synthesis:
The place of attack of the carbon atom by fluoride-anion is determined by the nature of its substituents and with increase of the spatial requirements the ketone formation ( for which this group stands directly at C=O to group) is took place.
Oxime of aldehydes and ketones are effective O-nucleophiles and are widely used in organic synthesis. Thus, oxyme acetone [72] and cyclic ketones [73] at the presence of a triethylamine form with perfluoro-2-methylpent-2-ene products of fluorine atom replacement at double bond and products of connection on it. At the same time, the reaction perfluoro-2-methyl-pent-2-ene with oxymes of aliphatic aldehydes resulted new fluorocontaining isoxazolines. The authors of [72-75] have found out a thermal rearrangement fluoro-alkenylic ethers oxymes to derivative pyrroline. We would like to point out the interest to heterocyclic compounds with perfluoroalkyl groups which show increased biological activity and can be employed in medicines and agriculture branches. As it has been shown [76], in comparison to acetone oxime, which reacts with perfluoro-2-methylpent-2-ene at the presence of the triethylamine, the reaction perfluoro-2-methylpent-2-ene with anti-oxyme acetophenone proceeds in the absence of the latter in a diglyme (or acetonitrile) with formation of a product of connection on double bond : 3- (2-trifluoromethylnonafluoro)-pentylic ether oxyme acetophenone. At the same time at the presence of a triethylamine the product of replacement of fluorine atom at internal double bond is 3-perfluoro-2-methylpentene-2-ylic ether oxyme acetophenone.
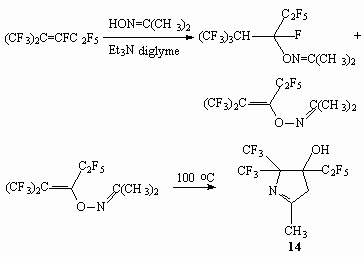
As opposed to terminal perfluoroolefins, which react with oxyme acetone producing only product of double bond connection, internal perfluoroolefins, for example perfluoro-2-methylpent-2-ene, issue mixture of products of connection and replacement of fluorine atom at double bond. And the yield of the latter product is increased with increasing of the basis amount. This is a stable compound when a storage at temperature not exceeding 30 oC, at 100 oC during 1 hour produce 4-oxy-3-methyl-5,5-bis(trifluoromethyl)-4-pentafluoroethylpyrroline-1 (14) [72].
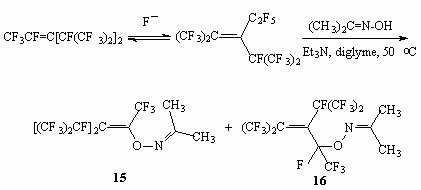
The interaction between trimers of a hexafluoropropylene with oxyme acetome yield two isomeric products (15) and (16) due to the nucleophilic fluorine replacement at double bond.
Other oxymes react with internal perfluoroolefins similarly. Thus, oxymes of cyclopentanone and cyclohexanone react with perfluoro-2-methylpent-2-ene at presence of catalitic amounts of bases, producing adducts and products of their dehydrofluorination (yield no more than 10 %) [73]. It is necessary to note, that vinylic ethers are unstable under heating and at 100 oC their complete transformation into pyrolidines is observed (17).
17 Reactions C, N-diphenylnitrone with octafluorobut-2-ene and octafluoronorborandiene at room temperature allow to obtain at once the products of a cyclization, i.e. corresponding isoxalidine [77].
2.2. The reaction of S-nucleophiles with internal perfluoroolefins The reactions of S-nucleophilic reagents with internal perfluoroolefins are rather sensitive to conditions of process realization, in particular temperatures. So, the reaction between tetrafluoroethylene pentamer and mercaptanes (at Et3N presence) results a mixture of products [78,79].
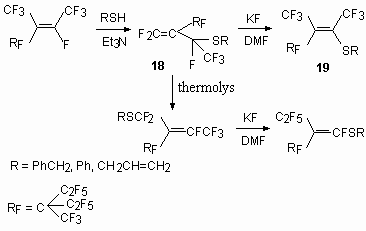
Thus a kinetically controllable product is obtained at temperatures –65 - -30 oC (18), whereas - thermodynamically controllable product is observed at room temperature (19) ( Table 1). Table 1. Temperature effect on the correlation products 18and 19 [78].
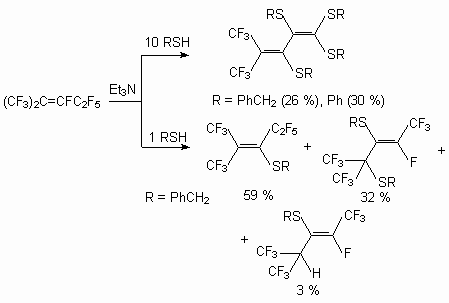
The reactions of hexafluoropropylene dimers with benzylmercaptane and thiophenol at the presence of a triethylamine yield a complex mixture of fluorinated vinylic sulfides with fluorine replacement at double bond and deeper transformation under fluoride-ions action take place [39]. First of all, it depends on the quantity of S-nucleophilic reagent used.
Trimers of a hexafluoropropylene react with mercaptanes at presence Et3N, forming derivative with SR substituents at double bond [49, 80].
Dialkyldithiocarbomates are used as S-nucleophilic agents [81].
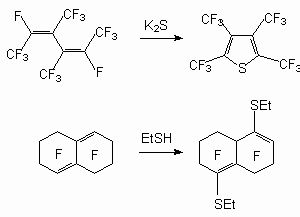
The examples from following articles illustrate these processes for other dienes: in the reaction between perfluoro-3,4-dimethylhex-2,4-diene and K2S a perfluorotetramethylthiophene was obtained [32], and when perfluoro-1,2,3,5,6,8a-hexahydronaphthalene reacted with ethylmercaptane 4,8-bis-ethylsulfanyldodecafluoro-1,2,3,5,6,8a-hexahydronaphthalene was produced [82].
to be continued |
Fluorine Notes, 2000, 12, 1-2
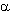 ,
, - trihydroperfluoroalcohols [27]. These reactions are
mainly in catalitic amounts of the basis, whereas their increase
results in the growth of the contents in reactionary mixtures of
non-limiting products - vinilic ethers. The use of Et3N
issues in the products of a fluorine replacement at internal double
bond, whereas in the presence of NaOH the product of two alcosilic
groups entry prevails.
- trihydroperfluoroalcohols [27]. These reactions are
mainly in catalitic amounts of the basis, whereas their increase
results in the growth of the contents in reactionary mixtures of
non-limiting products - vinilic ethers. The use of Et3N
issues in the products of a fluorine replacement at internal double
bond, whereas in the presence of NaOH the product of two alcosilic
groups entry prevails.
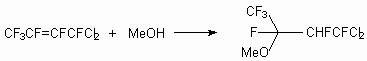
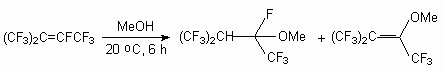
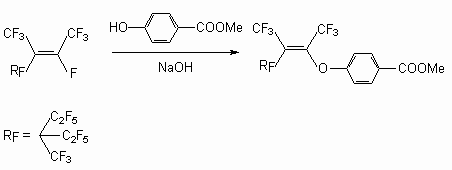
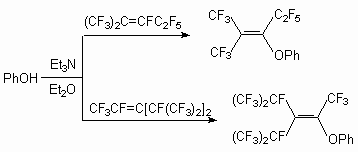
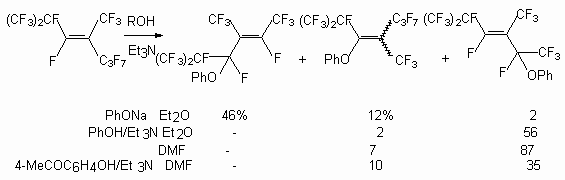
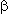 -position, yielding compound (6), whereas in case of phenols
the elimination of fluoride-anion occurs from
-position, yielding compound (6), whereas in case of phenols
the elimination of fluoride-anion occurs from