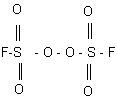Fluorine Notes, 2000, 12, 3-4
Reactions of fluoroolefins with participation of fluorine inorganic radical initiatorsVictor M. AndrushinReport 2. Peroxydisulfuryldifluoride - production and properties. A complex of properties that should be necessary for radical initiators of reactions of fluoroolefins have been formulated and grounded in report 1 . Peroxydisulfuryldifluoride most closely corresponds to the requirements and it has been chosen as a main radical reagent. Its main properties are given below. For the first time peroxydisulfuryldifluoride (PSF) was synthesized in 1957. It is peroxide of fluorosulfonic acid.
In contrast to majority of representatives of this class compounds, it is a stable compound without explosive properties. Its high stability is obviously explained by the symmetrical structure and influence of electronegative substituents at oxygens of the peroxide fragment of the molecule. Some tens of original research works have been devoted to the properties of PSF and its reactivity, but the values of various parameters presented by them differ considerably. We have taken the most commonly used values given in (1,2). PCF is a volatile liquid with b.p.67.1oC and melting point of 55.4oC, the density of liquid PSF at 20oC is 1.680g/cm, energy of the O-O bond dissociation is 22,4 kcal/mole. The 19F NMR, and IR- spectra for PSF have been studied in detail. The characteristics of the fluorosulfate radical including the IR-spectrum , absorption spectra in visible region and fluorescence under laser action have been investigated also. Peroxydisulfuryldifluoride is thermally stable up to 400oC, insensible to mechanical actions (impact, friction), phase transformations, open flame. In contrast to other covalent fluorosulfates that vigorously, sometimes with explosion react with even moisture traces, PSF is hydrolyzed slowly. That allows carrying out its purification from admixtures of other fluorosulfates and fluorosulfonic acid by a short term treatment with water at a temperature of 10-15oC maximum and vigorous shaking. PSF is not mixed with water and can be separated from water in a common separating funnel. PSF solutions in fluorosulfonic acid are not conductive that obviously confirms a radical character of the molecule dissociation. At the same time PSF is a highly reactive compound. It readily reacts with halogens, many different elements, their oxides, salts, hydrides and other derivatives. In literature one can find descriptions of the reactions of PSF with sulfur, selenium, tellurium, antimony, ammonia, hydrogen sulfide, nitrozyl- and nitrile chlorides, oxyhalides of phosphorus, sulfur and tellurium; isocyanates, phosphorus fluorides, sulfur fluorides, metal carbonyls, nitrogen fluorides, oxyhaloids, other fluoroperoxides etc. In dependence on the reagent nature PSF behaves either as oxidizer or forms addition products. Some reactions, for example with carbon and sulfur oxides, nitrogen and sulfur fluorides and some others have been studied kinetically. The PSF reactions with organic compounds are presented as well. In alkanes including substituted ones PSF replaces hydrogen atoms to form appropriate fluorosulfonates according to the following scheme: RH + S2O6F2 In this case the appropriate poly(fluorosulfato) derivatives can be produced also. Hydrogen can be substituted most readily in alkanes, chladones and no substitution was observed in amines, hydroxyls, bromoalkanes. Chlorine atoms are rather readily replaced with fluorosulfate group. The high reactivity of PSF allows replacing hydrogen with fluorosulfate group even in so inert compounds as monohydroperhaloid alkanes. The substitution reaction of secondary hydrogen atoms proceeds more readily. A.V.Fokin, Yu.N.Studkov et al. have determined that heminal bis(fluorosulfato) derivatives are rather unstable and can destroy according to the scheme: R(OSO2F)2 to form the appropriate oxides and ketones. In all, some tens of such reactions have been described and their radical character is beyond any doubt. In some cases decomposition of the produced compounds takes place. The reaction of PSF with salts proceeds in a similar way as hydrogen substitution. In comparison with the above reaction of hydrogen replacement, substitution of haloids in haloidalkanes and haloidanhydrides of acids proceeds more smoothly. The reactivity of PSF in these reactions increases considerably in the presence of bromine or fluorosulfonic acid. In the first case more reactive bromine fluorosulfate is formed whereas in the second case polarization of the PSF molecule occurs. The reaction of PSF with alcohols including fluorinated ones proceeds with destruction whereas their alcoholates allow to obtain the appropriate peroxides. The interaction with perfluorocarbonic acids did not reveal similar peroxides. In amines and amides of perfluorocarbonic acids hydrogen substitution does not meet any difficulties though N-fluorosulfates are produced in a low yield. PSF with organofluoric peroxides forms mixed peroxide derivatives that can be produced by the interaction of PSF with anhydrides of perfluoroacids. The investigation of the interaction of PSF with unsaturated compounds is of particular interest for our study. In benzene and toluene only substitution of hydrogen atoms in the ring takes place and no addition along the multiple bond was discovered. The addition along the C-C multiple bonds have been studied on many examples beginning from the 60-s. In the interactions with tetrafluoroethylene, chlorotrifluoroethylene, perfluorocyclopentene, hexafluoropropylene, perfluorobutene, perfluorocyclobutene, trichloroethylene, vinyl fluoride and vinyl chloride etc the addition of the fluorosulfate radicals along the double bond takes place to form vicinal bis(fluorosulfate) derivatives. The reaction with perfluorobenzene is more specific: a mixture of 1,4-bis(fluorosulfate)- and 1,2,3,4-tetra(fluorosulfate)-derivatives is formed. The addition reaction has been investigated completely enough and its radical nature is beyond any doubt. The addition along the C-N multiple bonds in diazadienes proceeds under mild conditions and the addition does not occur along the single C=N bonds, it is followed by substitution of functional groups with fluorosulfate ones, for example in pentafluoroguanidine. Complete saturation of the triple bond takes place in nitriles, in imines the addition is followed by oxidizing dimerization of an imine with further addition of PSF to the forming diazodiene. Thus, the literature data analysis confirms mainly the radical character of the PSF reactions. We have determined the EPR spectrum of SO3F radical on a RE 1306 spectrometer taken CuCl *2H20 as the standard. The spectrum of the SO3F radical at 196oC is given in Fig.
At room temperature the average radical concentration was 2.13*10 16 1/g( 5.2*10 18 1/mole). As it is seen from the experimental data, the radical concentration is sufficient enough for confirmation of the radical character of the described reactions. There are some methods of the PSF synthesis in literature. The catalytic and photochemical fluorination of sulfur anhydride is presented most widely but in this case explosive fluorine fluorosulfate is formed. PSF can be produced from fluorine fluorosulfate by its treatment with sulfuric anhydride and from chlorine fluorosulfate under exposure to UV radiation. The electrolysis of HSO3F *KSO3F melt is of technical interest, its experimental examination has shown that the yield of the target product did not exceed 60% on platinum electrodes in a glass cell that is suitable enough for making laboratory samples. For the use on a large scale thorough knowledge of the process is necessary. We consider the reaction between chlorine fluorides and sulfuric anhydride as the most convenient method to produce PSF. Monofluoride gives chlorine fluorosulfate which then is irradiates with simultaneous chlorine sampling. But one should know that some kinds of quartz are unstable in the mentioned media. The interaction of chlorine trifluoride with sulfuric anhydride at room temperature gives a stoichiometric mixture of PSF and chlorine fluorosulfate that is readily separated on a rectification column (b.p.45.1oC and 67.1oC respectively) In the reaction of chlorine pentafluoride with sulfuric anhydride PSF is not formed. We have not used other methods to synthesize PSF though fluorination of sulfuric anhydride with fluorosulfonic acid, fluorides of xenon, chromine and uranium and trifluoromethylhypofluorite is of a certain interest as well. We believe that PSF is an available enough product for laboratories and researchers having operational experience in work with fluorine and inorganic fluorides. |
Fluorine Notes, 2000, 12, 3-4