Fluorine Notes, 1998, 1, 7-8
Influence of the structure of a layer of surfactant adsorbed on a solid surface on interaction of lubricant with the surface of parts of friction units.
V.V. Amfeeteatrov, L.E. Vestfal, V.E. Moungalov, S.V.Vershilov, E.V. Ireesova
A task to reduce friction and wear of friction parts remains actual due to a constant growth of energy saturation of machines and mechanisms .
One of the ways of its solution is expansion of the range of liquid and boundary friction to the direction of greater loadings by means of applying various antiscuff and antiwear additions to lubricants.
In this connection it seems reasonable to test a possibility to use some fluorine-containing surfactants as anti-wear additions to oils, as well as for preliminary treatment of the surface of parts of friction units by them.
2-perfluoroalkyl-5-mercaptotriazole has been chosen as a surfactant.
The liquid and boundary friction is determined by the interaction of the lubricant with the surface, hence the effect of the surfactant layer structure on this surface was studied.
The study was carried out by a method of the "oscillating needle"(1) assuming that the process parameters (logarithmic damping decrement D and frequency f ) of free rotary oscillations of a pendulum instrument needle oil-immersed will change only in the case of surfactant layer applying on the needle surface. f = 1/T, A = ln An/An+1 , where T is the period, An and A n+1 are the amplitudes of the two neighboring oscillations, all of them are the registrated parameters of the free oscillations process.
The surfactant was applied on the needle surface (steel with Cr 18%, Ni –10%, Ti-1%) by means of adsorption from solutions of different concentration (from 0.001 % to 0.01 %) see C % in the Table.
Automobile oil was used as a lubricant.
The parameters f and D, for the surfactant layer absorbed from the solutions of different concentration on the needle surface are given in the Table.
TABLE
|
C,% |
f*102, hz |
D / p |
fk2*104 =G’ |
tg d |
D / p *f*100= m |
|
0 |
0.90 |
0,235 |
0,040 |
4.78 |
- |
|
0,001 |
0,99 , |
0,173 |
0,22 |
0,76 |
- |
|
0,005 |
1,32 |
0,247 |
0,96 |
0,45 |
- |
|
0.006 |
1,81 |
0,296 |
2,45 |
0,39 |
- |
|
0,008 |
1,02 |
0,359 |
0,27 |
1,41 |
- |
|
0,01 |
0,82 |
0,025 |
- |
- |
0,021 |
As it is seen from the Table, the increase in f for the surfactant layer absorbed from the solution of the minimum concentration (0.001%) in comparison with a couple "steel-oil" points out that the surfactant layer possesses elastic properties due to the structure in the form of Langmuir " paling" filled with oil.
Viscoelastic properties of materials are characterized by elastic component of shear modulus G' and mechanical losses factor tg d . These parameters for the case of free shear oscillations of the material are defined by the following relations:
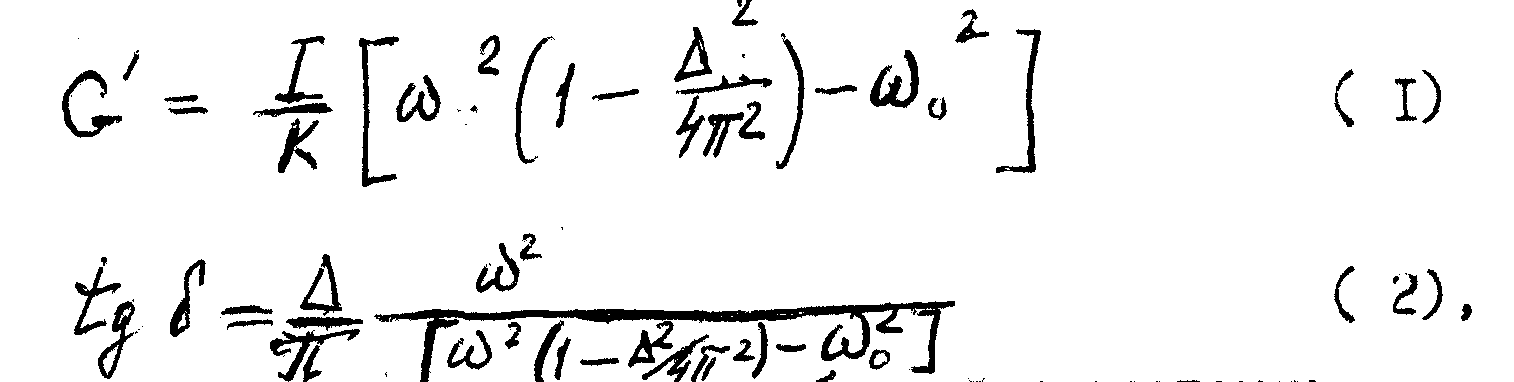
where I is the moment of inertia of the oscillating system,
K is form factor;
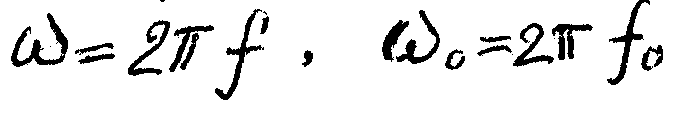
where fo is the natural frequency of free oscillations of the pendulum instrument.
Since the thickness of the surfactant layer is unknown, in the Table there are given the values proportional to G' and m ,
An increase in elasticity of the surfactant layer and reduction of the mechanical losses in it with an increase in the surfactant solution concentration is the evidence of the "paling" thickness (density) increase . Nonlinear character of the change in the layer elastic properties (Fig.1) along with the increase of the "paling"density is similar to the nonlinear growth of the elasticity of composite materials (in particular reinforced ones) with substantially different properties of the binder and reinforcing component ( the binder has a low value of the shear modulus and the reinforcing component has a high one).
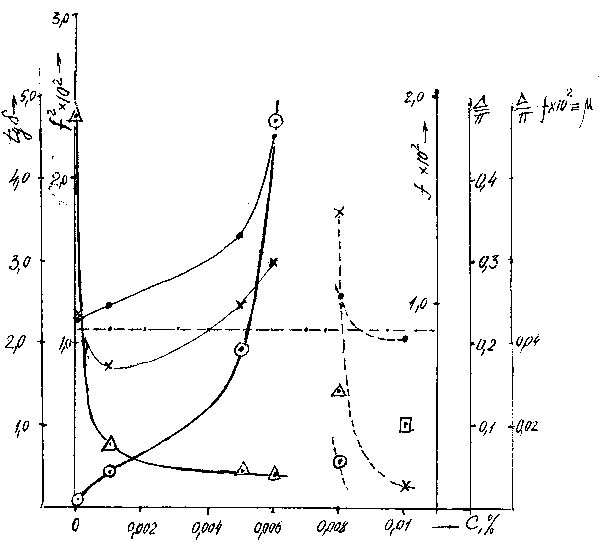
Picture1. Dependence of the parameters of the surfactant layer structure on the solution concentration
| |
|
| ----the most possible behavior of the dependence | |
A diagram representing oil interaction with the solid surface and dependences on the structure of the surfactant layer with different density of "paling" is given in Fig.2.
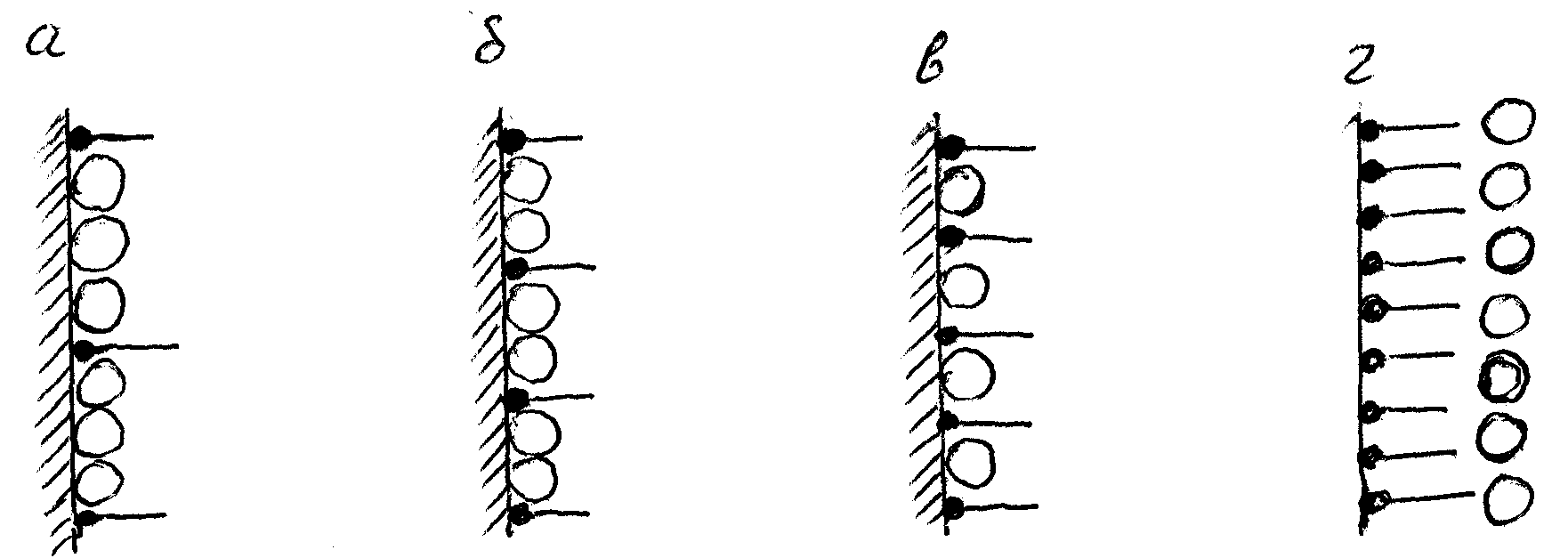
where O-is an oil molecule ,
a)- surfactant layer from a solution of low concentration
b),c)- elevated concentration
d) - high concentration
When the concentration of the surfactant solution is 0.01 % , f becomes lower than fo, D / p becomes an order of magnitude lower than the value of the couple "steel-oil". It is the evidence of no interaction with the needle surface because of the density of "paling" has overcame the critical value. As a consequence of mosaic character of active centers distribution on the solid surface, such a structure of the surfactant layer appears notjumpwise, this is confirmed by a presence of the right slope of the dependences of f and D / p on the surfactant solution concentration.
The exposed dependences of influence of the surfactant layer structure on the solid surface wetting with oil have the following applications:
1. Applying surfactant layers with Langmuir "paling" of maximum density ( but lower than the critical value) is reasonable for processing friction parts of heavy-duty friction units operating under the conditions of liquid friction. In our case such a layer is formed from the surfactant solution of ac. 0.006% concentration.
2. For friction units operating under the conditions of boundary friction it seems reasonable to apply fluorosurfactant layers with dense "paling" avoiding the oil contact with the solid surface. It will allow to take away the requirement of high lubricating properties of instrumental oils.
CONCLUSIONS.
The interaction of the lubricant with the solid surface with a monomolecular layer of the fluorosurfactant applied on it changes qualitively at the critical density of Langmuir "paling" of the film: from oleophilic character at the "paling"density below the critical value to oleophobic character at the "paling"density above the critical value.
This effect of the surfactant film structure on interaction of the oil with the solid surface should be taken into account when these layers are formed on the surfaces of friction units of mechanisms of different purposes.
LITERATURE
1. Cumper C.W., Alexander A.E., Trans. Faraday Soc., N 46, 235, 1950
Fluorine Notes, 1998, 1, 7-8
